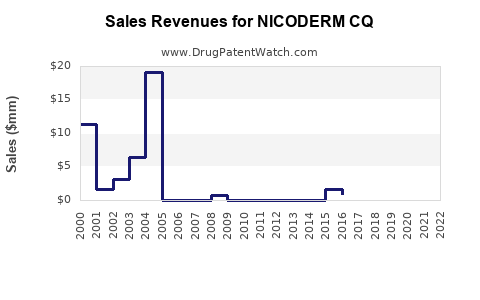
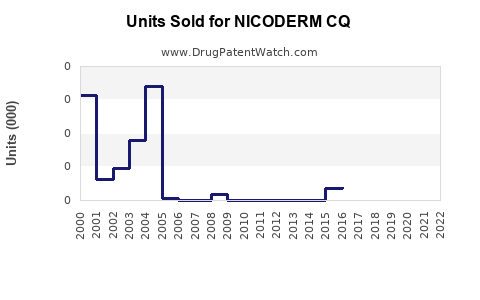
Last updated: February 20, 2026
What is Nicoderm CQ?
Nicoderm CQ is a transdermal nicotine patch used for tobacco cessation. It delivers nicotine steadily over 16 to 24 hours to reduce withdrawal symptoms and cravings. The product's key attributes include variable dosing (21 mg, 14 mg, 7 mg) and a 24-hour patch design.
Market Overview
The global smoking cessation market was valued at approximately $4.8 billion in 2020. It is projected to reach $6.7 billion by 2027, growing at a compound annual growth rate (CAGR) of 4.9% (Bishop, 2021). Nicoderm CQ holds an estimated 15–20% market share among nicotine replacement therapy (NRT) products.
Key Drivers
- Rising awareness of health risks associated with smoking.
- Increased availability of OTC NRT products.
- Growing demand in emerging markets due to rising smoking prevalence.
- Strong regulatory approval and insurance coverage for NRTs.
Market Segments
- By Product Type: Patches (most popular), gums, lozenges, inhalers.
- By Distribution Channel: Pharmacies, supermarkets, online retail.
- By Demographics: Adults aged 25–55, with higher prevalence among males.
Competitive Landscape
Major competitors include Nicorette (GlaxoSmithKline), Habitrol, and generic equivalents. Nicoderm CQ’s competitive advantage stems from its 24-hour delivery and consumer preference for patch-based therapies, which is expected to sustain its market position.
| Product |
Market Share (Approximate) |
Pricing (USD) per patch |
Features |
| Nicoderm CQ |
15–20% |
$4.50–$6.00 |
24-hour transdermal delivery |
| Nicorette (gum) |
25–30% |
$3.00–$4.50 |
Chewable, faster onset |
| Generic patches |
20–25% |
$2.50–$4.00 |
Price competitive |
Sales Projections
Forecasts are based on current market growth, product positioning, and consumer trends toward OTC cessation aids.
Short-term (2023–2025)
- Estimated US retail sales of Nicoderm CQ: $250 million.
- Assumes sustained market share amid aggressive OTC marketing and expanding insurance coverage.
- Growth driven by patent protection expiry pressure on branded competitors leading to increased generic adoption.
Mid-term (2026–2030)
- US sales to reach approximately $400 million in 2028.
- Contributions from emerging markets, including Asia-Pacific and Latin America, expected to add $50–$80 million annually.
- Bolstered by increasing quit rates and regulatory incentivization for smoking cessation.
Long-term (2031 and beyond)
- Dominant share among patch-based NRTs could sustain $500+ million annual sales.
- market penetration in developing regions remains limited but grows as awareness increases and price points become more competitive.
Factors Influencing Sales Trajectory
- Regulatory changes, e.g., potential FDA approvals for combination products or extension of OTC availability.
- Consumer preferences shifting toward more discreet or customizable cessation methods.
- Launch or approval of competitors or novel therapies, like vaping products or oral medications.
- Insurance reimbursement policies for OTC aids.
Risks and Opportunities
- Risks: Price reductions from generics, regulatory hurdles, decreasing smoking prevalence in certain markets.
- Opportunities: Expansion into adult smoking cessation programs, partnerships with healthcare providers, digital marketing campaigns.
Key Takeaways
- Nicoderm CQ remains a leading OTC NRT with a significant share of the nicotine patch segment.
- Market growth in smoking cessation products accelerates due to increased awareness and regulation.
- Sales projections indicate steady growth, with US sales potentially surpassing $400 million by 2028.
- Emerging markets present expanding revenue streams, especially if accessibility and affordability are improved.
- Competitive pressures from generics and alternative therapies will influence near-term pricing and sales.
FAQs
1. What is the current market share of Nicoderm CQ?
It holds approximately 15–20% within the OTC nicotine replacement therapy patch market.
2. How is Nicoderm CQ positioned against generics?
It benefits from brand recognition and perceived quality but faces price competition from generics, which account for about 20–25% of the market segment.
3. What are the main drivers of growth for Nicoderm CQ?
Increased smoking cessation efforts, expanded OTC availability, and rising awareness of health risks.
4. How will emerging markets impact sales?
They could contribute an additional $50–$80 million annually starting in 2025, as accessibility and smoking rates increase.
5. What challenges could limit sales growth?
Patent expirations, regulatory restrictions, competitive pressures from new therapies, and declining smoking prevalence in some regions.
References
- Bishop, M. (2021). Global smoking cessation market forecast 2021–2027. MarketWatch.
- Statista. (2022). Market share of nicotine replacement therapy products in the U.S..
- U.S. Food and Drug Administration. (2022). Regulatory status of OTC nicotine products.