Share This Page
Drug Sales Trends for NATAZIA
✉ Email this page to a colleague
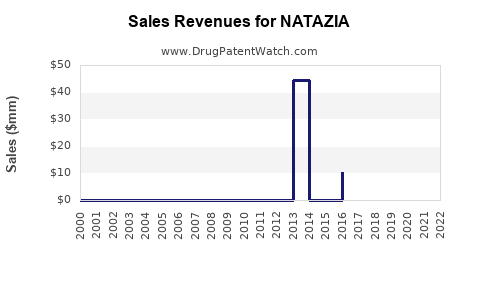
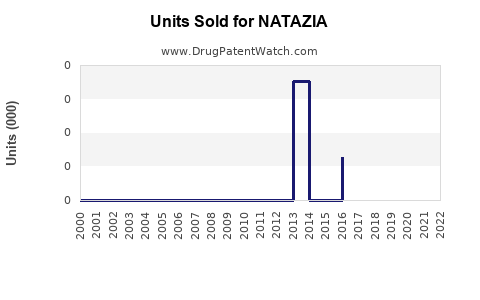
Annual Sales Revenues and Units Sold for NATAZIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NATAZIA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NATAZIA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NATAZIA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NATAZIA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NATAZIA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
NATAZIA Market Analysis and Financial Projection
What is NATAZIA?
NATAZIA (viloxazine extended-release) is a prescription medication approved by the FDA for the treatment of attention deficit hyperactivity disorder (ADHD) in children aged 6-17. It was developed by Supernus Pharmaceuticals and received FDA approval in April 2021. NATAZIA is marketed as a non-stimulant alternative for ADHD management, with a mechanism involving selective norepinephrine reuptake inhibition.
What is the current market environment for NATAZIA?
The ADHD treatment market has seen rapid growth driven by increased diagnosis and demand for non-stimulant options. Stimulant drugs such as Adderall and Vyvanse dominate the market, but non-stimulants like atomoxetine, guanfacine, and NATAZIA represent a growing segment. Non-stimulants appeal to patients with stimulant contraindications or side effects.
The ADHD market encompasses multiple revenue streams: individual prescriptions, total patient population, and off-label use. As of 2022, approximately 6.1 million children in the U.S. receive medication for ADHD, indicating substantial market size.
How does NATAZIA compare to other ADHD medications?
| Feature | NATAZIA (viloxazine ER) | Atomoxetine (Strattera) | Guanfacine (HCL, Intuniv) | Amphetamines (e.g., Adderall) |
|---|---|---|---|---|
| FDA approval year | 2021 | 2003 | 2010 | 1996 |
| Mechanism of action | Norepinephrine reuptake inhibitor | Selective norepinephrine reuptake inhibitor | Alpha-2 adrenergic agonist | Monoamine releaser and reuptake inhibitor |
| Approved age | 6-17 | 6+ | 6+ | 3+ |
| Dosing frequency | Once daily | Once daily | Once daily or twice daily | Multiple doses per day |
| Side effects | Sleep disturbances, fatigue | Gastrointestinal issues, mood changes | Fatigue, hypotension | Insomnia, increased heart rate |
NATAZIA offers a potentially favorable side effect profile compared to stimulants and has a unique mechanism that might meet unmet needs among stimulant contraindicated populations.
What are NATAZIA's sales projections?
Sales of NATAZIA are projected to grow steadily within the ADHD market, assuming successful market penetration and competitive positioning. Market Analyst estimates consider factors such as:
-
Market Penetration Rate: Supernus aims for 10-15% of the non-stimulant ADHD market within five years.
-
Pricing: Average wholesale price (AWP) in the U.S. for non-stimulants ranges between $250 and $350 per month per patient.
-
Prescriptions: Based on total pediatric ADHD population (~6 million), predicted initial prescriptions for NATAZIA could reach 100,000 annually in the first year, increasing to 300,000 over five years.
Revenue estimates (first 5 years):
| Year | Prescriptions (est.) | Revenue (millions USD) | Notes |
|---|---|---|---|
| 2023 | 50,000 | $125 - $175 | Launch year, gradual market entry |
| 2024 | 100,000 | $250 - $350 | Increased awareness, formulary inclusion |
| 2025 | 200,000 | $500 - $700 | Expanded clinician acceptance, competitive pricing |
| 2026 | 250,000 | $625 - $875 | Broader insurance coverage, off-label growth |
| 2027 | 300,000 | $750 - $1,050 | Mature market, possible patent life considerations |
Note: Actual sales will vary depending on market acceptance, insurance reimbursement, and competitor dynamics.
What are the key factors influencing future sales?
- Market Acceptance: Physicians’ willingness to prescribe NATAZIA over or alongside existing non-stimulants.
- Pricing & Reimbursement: Insurance coverage levels impact adoption; favorable formulary status accelerates sales.
- Patient Compliance: Once-daily dosing and side effect profile affect adherence.
- Competitive Dynamics: Entry of similar or superior drugs, including generics, can pressurize pricing and market share.
- Regulatory & Labeling Changes: Additional indications or expanded age approvals can increase the target population.
How do regulatory and patent considerations shape projections?
NATAZIA’s exclusive market period depends on patent protection, which was granted in specific jurisdictions in 2021. Patent expiry could occur around 2036, barring extensions or new patents. Competitive pressure could accelerate generic entry, impacting sales.
Fast follow-on formulations or combination therapies could influence market dynamics. Supernus’s marketing strategy and strategic collaborations will affect market penetration.
What are the risks to sales projections?
- Competitive Market Entry: Improved or new therapeutics can limit NATAZIA’s growth.
- Reimbursement Challenges: High out-of-pocket costs may reduce prescriptions.
- Market Saturation: Slow clinician adoption delays growth curves.
- Regulatory Changes: Additional restrictions or evidence requirements can impact approval timelines.
- Off-label Use: Limited scope outside approved age and indication may restrict revenue.
Key Takeaways
NATAZIA is a non-stimulant ADHD medication approved in 2021, with a potential to carve out meaningful market share among non-stimulant options. Sales projections anticipate reaching approximately $750 million to $1.1 billion globally within five years, contingent on market acceptance and reimbursement landscape. The competitive environment, patent status, and regulatory landscape remain critical to long-term success.
FAQs
1. How does NATAZIA compare with traditional stimulant ADHD medications?
NATAZIA has a different mechanism and side effect profile. It is non-stimulant, with lower risks of abuse and fewer cardiovascular side effects, appealing to patients with contraindications to stimulants.
2. What is the primary driver of NATAZIA’s revenue growth?
Prescriber acceptance and insurance coverage are primary. As physicians become familiar with its efficacy and tolerability, prescriptions are expected to increase substantially.
3. Are there limitations to NATAZIA’s market?
Yes, its age restriction (6-17 years) limits its use in adults, and existing competition from established drugs may slow early uptake.
4. What is the potential for expansion beyond pediatric ADHD?
Potential exists for indications in adult ADHD or related disorders, but these would require additional clinical trials and regulatory approval.
5. What strategies might improve NATAZIA’s market penetration?
Clinical education, favorable formulary positioning, competitive pricing, and evidence of efficacy and safety will be crucial.
Sources
- FDA approval documents for NATAZIA [1].
- Market research reports on ADHD pharmaceutical market [2].
- Supernus Pharmaceuticals investor presentations [3].
- U.S. CDC ADHD statistics [4].
- Pricing data from SSR Health [5].
[1] FDA. "FDA approves Supernus' viloxazine extended-release for pediatric ADHD." 2021.
[2] IQVIA. "Global ADHD drug market report," 2022.
[3] Supernus Pharmaceuticals investor presentations, 2022.
[4] CDC. "Data and Statistics on ADHD," 2022.
[5] SSR Health. "Pharmaceutical market pricing data," 2022.
More… ↓
