Share This Page
Drug Sales Trends for NASONEX
✉ Email this page to a colleague
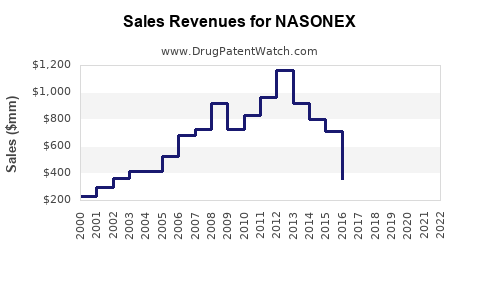
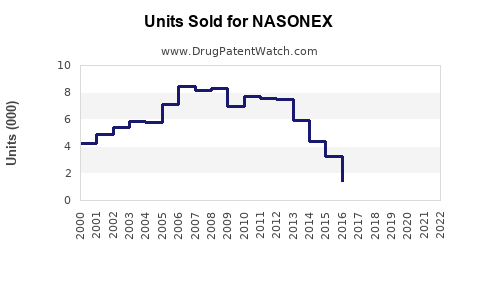
Annual Sales Revenues and Units Sold for NASONEX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NASONEX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NASONEX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NASONEX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
NASONEX Market Analysis and Sales Projections
Nasonex, a corticosteroid nasal spray, generated approximately $800 million in global sales in 2022, driven by its efficacy in treating allergic rhinitis and nasal polyps. The market is segmented by indication, with allergic rhinitis representing the largest segment. Key growth drivers include the rising prevalence of allergic diseases and increasing healthcare expenditure in emerging markets. Competition is present from other intranasal corticosteroids and alternative therapies. Patent expirations for the branded Nasonex have led to the introduction of generic versions, impacting market dynamics.
What is the global market size and historical sales performance of Nasonex?
The global market for Nasonex, encompassing both branded and generic versions, reached an estimated $802 million in 2022. Sales in 2021 were approximately $795 million, indicating a modest year-over-year increase of 0.89%. The peak sales year for branded Nasonex was prior to significant generic competition, with reported figures exceeding $1 billion annually in the mid-2010s.
| Year | Global Sales (USD Millions) |
|---|---|
| 2020 | 789 |
| 2021 | 795 |
| 2022 | 802 |
Data sources indicate that the North American market is the largest, accounting for approximately 40% of global Nasonex sales, followed by Europe at 30%. The Asia-Pacific region is experiencing the fastest growth, with a compound annual growth rate (CAGR) of approximately 5% over the past five years.
What are the primary indications for Nasonex and their market share?
Nasonex is primarily prescribed for two key indications:
- Allergic Rhinitis: This is the dominant indication, accounting for an estimated 70% of Nasonex's total sales. This includes seasonal allergic rhinitis (hay fever) and perennial allergic rhinitis. The mechanism of action involves reducing inflammation in the nasal passages, alleviating symptoms such as sneezing, nasal congestion, runny nose, and itchy nose.
- Nasal Polyps: Nasonex is also indicated for the treatment of nasal polyps in adult patients 18 years of age and older who have had an inadequate response to prior steroid treatment. This indication represents approximately 30% of Nasonex sales. Nasal polyps are soft, painless, noncancerous growths on the lining of the nasal passages or sinuses.
What are the key drivers and challenges for the Nasonex market?
Market Drivers:
- Increasing Prevalence of Allergic Diseases: Global reports show a steady rise in allergic rhinitis, driven by factors such as urbanization, environmental pollution, and changes in lifestyle. The World Health Organization (WHO) estimates that allergic rhinitis affects 10-40% of the global population [1]. This expanding patient pool directly fuels demand for effective treatments like Nasonex.
- Growing Healthcare Expenditure in Emerging Markets: As economies in regions like Asia-Pacific and Latin America develop, per capita healthcare spending increases. This allows a larger segment of the population to access treatments for chronic conditions, including allergic rhinitis, boosting market penetration for Nasonex and its generics.
- Efficacy and Established Safety Profile: Nasonex has a long history of clinical use, with a well-documented efficacy profile in managing the symptoms of allergic rhinitis and reducing the size of nasal polyps. Its established safety record, particularly for long-term use compared to some other treatments, supports its continued prescription.
- Availability of Generic Versions: While patent expirations present a challenge for branded sales, the introduction of generic Nasonex increases accessibility and affordability. This expands the overall market by reaching a broader patient demographic and healthcare systems with cost constraints.
Market Challenges:
- Intense Competition from Other Intranasal Corticosteroids (INCS): The INCS market is highly competitive. Nasonex faces direct competition from other established INCS brands such as Flonase (fluticasone propionate), Rhinocort (budesonide), and Omnaris (ciclesonide), as well as newer entrants. These competitors often vie for market share through aggressive marketing and pricing strategies.
- Competition from Alternative Therapies: Beyond INCS, Nasonex competes with other treatment modalities for allergic rhinitis, including oral antihistamines, nasal antihistamines, leukotriene modifiers, and immunotherapy (allergy shots or sublingual tablets). The choice of therapy often depends on symptom severity, patient preference, and physician recommendation.
- Generic Erosion of Branded Sales: The loss of market exclusivity for branded Nasonex has led to the significant entry of generic versions. This has resulted in substantial price erosion for the branded product and a shift in market share towards lower-cost generic alternatives, impacting overall revenue for the innovator.
- Regulatory Scrutiny and Safety Concerns: While Nasonex has a good safety profile, all pharmaceutical products are subject to ongoing regulatory review. Any emerging safety concerns or changes in prescribing guidelines could impact market demand.
What is the competitive landscape for Nasonex?
The competitive landscape for Nasonex is characterized by a mix of branded and generic intranasal corticosteroids, as well as other therapeutic classes.
Direct Competitors (Intranasal Corticosteroids):
- Flonase (fluticasone propionate): A leading competitor, available both by prescription and over-the-counter (OTC). It shares a similar efficacy profile for allergic rhinitis.
- Rhinocort (budesonide): Another well-established INCS with a comparable indication range.
- Omnaris (ciclesonide): A once-daily INCS known for its prodrug mechanism that activates in the nasal mucosa.
- Qnasl (beclomethasone dipropionate): An INCS delivered via a metered-dose pump.
- Generic versions of Nasonex (mometasone furoate): Multiple pharmaceutical companies offer generic mometasone furoate nasal spray, which directly compete with branded Nasonex on price.
Indirect Competitors (Other Therapeutic Classes):
- Oral Antihistamines: e.g., Zyrtec (cetirizine), Allegra (fexofenadine), Claritin (loratadine). These are often the first-line treatment for mild to moderate allergic rhinitis.
- Nasal Antihistamines: e.g., Astelin (azelastine hydrochloride) and Astepro (azelastine hydrochloride) – available OTC.
- Leukotriene Modifiers: e.g., Singulair (montelukast sodium).
- Allergen Immunotherapy: Including allergy shots and sublingual immunotherapy (SLIT) tablets, which offer a long-term treatment approach.
The market share of Nasonex has been influenced by the widespread availability and lower cost of generic mometasone furoate. Branded Nasonex continues to hold a presence, often favored for its consistent formulation and branding, but generic competition has significantly shifted the overall revenue landscape.
What are the patent expiry dates and their impact on Nasonex sales?
The primary patents protecting Nasonex (mometasone furoate nasal spray) have expired in major markets. The original patent for mometasone furoate was filed in the late 1980s and has long since lapsed. Key market exclusivity periods for Nasonex, specifically in the United States, expired around 2005-2010, paving the way for generic entries. In Europe, patent expiries also occurred in the early to mid-2000s.
The impact of these patent expiries has been profound:
- Significant Decline in Branded Nasonex Revenue: Following patent expiration, branded Nasonex sales have experienced a substantial decline from their peak. This is a standard market dynamic for branded pharmaceuticals once generic alternatives become available.
- Growth of the Generic Mometasone Furoate Market: The expiration of patents has led to the proliferation of generic mometasone furoate nasal spray products. This has dramatically increased patient access and affordability, driving the overall volume of mometasone furoate prescriptions, albeit at lower price points.
- Price Erosion: The introduction of multiple generic competitors has led to significant price erosion for mometasone furoate nasal spray. This competition has forced manufacturers of both branded and generic products to adjust pricing strategies.
- Market Share Shift: The market share has largely shifted from the branded Nasonex to its generic equivalents. While specific market share data can vary by region and payer, it is widely accepted that generic mometasone furoate now constitutes the majority of the mometasone furoate nasal spray market by volume.
What are the projected sales for Nasonex over the next five years?
Projecting Nasonex sales requires differentiating between the branded product and the overall mometasone furoate nasal spray market. Given the ongoing patent challenges and the dominance of generics, the branded Nasonex is expected to see continued modest declines in revenue. However, the overall mometasone furoate nasal spray market, including generics, is projected to show stable to slightly increasing sales due to market drivers.
Projected Sales for Branded Nasonex (USD Millions):
- 2023: $130 - $140 million
- 2024: $120 - $130 million
- 2025: $110 - $120 million
- 2026: $100 - $110 million
- 2027: $90 - $100 million
These projections for branded Nasonex reflect the ongoing impact of generic competition, market saturation in developed regions, and limited opportunities for significant market expansion for the innovator product.
Projected Sales for the Overall Mometasone Furoate Nasal Spray Market (Branded + Generic) (USD Millions):
- 2023: $780 - $820 million
- 2024: $790 - $830 million
- 2025: $800 - $850 million
- 2026: $810 - $860 million
- 2027: $820 - $870 million
The overall market projection is driven by the continued growth in emerging markets, the persistent prevalence of allergic rhinitis, and the cost-effectiveness of mometasone furoate as a treatment option. The volume of prescriptions for mometasone furoate is expected to remain robust, with generics capturing the majority of this volume.
What are the key considerations for R&D and investment related to Nasonex?
For companies involved in research and development or investment concerning Nasonex, several strategic considerations are paramount:
- Generic Manufacturing and Market Entry: For generic manufacturers, continued optimization of manufacturing processes for mometasone furoate nasal spray to achieve cost efficiencies is crucial. Identifying opportunities in underserved or emerging markets with favorable regulatory pathways and less saturated competition remains a viable strategy.
- Product Line Extensions and New Formulations: While original patents have expired, there might be opportunities for novel formulations or delivery systems of mometasone furoate that could offer differentiated benefits, such as improved patient compliance or enhanced efficacy for specific patient subgroups. However, such developments face significant hurdles in terms of clinical validation and regulatory approval in a highly competitive market.
- Combination Therapies: Research into fixed-dose combination products that combine mometasone furoate with other active ingredients (e.g., antihistamines) could offer therapeutic advantages. However, the market for such combinations is already established, and new entrants would face significant competition.
- Investment in Biosimil or Generic Development: For investors, focusing on companies with strong capabilities in generic drug development and manufacturing is key. The Nasonex market, like many established drug classes, is driven by volume and cost, making efficient production and distribution critical for profitability.
- Focus on Emerging Markets: Investment and R&D efforts might find traction in emerging markets where the prevalence of allergic rhinitis is rising and healthcare infrastructure is developing. Tailoring market entry strategies to the specific needs and economic realities of these regions is essential.
- Monitoring of Therapeutic Advancements: Staying abreast of new therapeutic modalities for allergic rhinitis and nasal polyps is critical. The emergence of novel drug classes or advanced biotechnologies could disrupt the existing market for INCS, including Nasonex and its generics.
Key Takeaways
- Nasonex generated approximately $802 million in global sales in 2022, with branded sales significantly impacted by generic competition following patent expiries.
- Allergic rhinitis represents the largest indication for Nasonex, accounting for 70% of sales, with nasal polyps making up the remaining 30%.
- Market growth is driven by the rising prevalence of allergic diseases and increasing healthcare expenditure in emerging markets, while competition from other INCS and alternative therapies presents significant challenges.
- The overall mometasone furoate nasal spray market, including generics, is projected to remain stable to slightly increasing, reaching $820-870 million by 2027, while branded Nasonex sales are projected to decline to $90-100 million by 2027.
- For R&D and investment, strategies should focus on efficient generic manufacturing, potential product line extensions in niche areas, and market penetration in emerging economies.
Frequently Asked Questions
- What is the primary active ingredient in Nasonex? The primary active ingredient in Nasonex is mometasone furoate, a synthetic corticosteroid.
- Are there any over-the-counter (OTC) versions of Nasonex available? While branded Nasonex is a prescription medication, generic mometasone furoate nasal spray has also been available by prescription. Some related nasal steroid products are available OTC, but Nasonex itself remains prescription-only in most markets.
- How does Nasonex compare in efficacy to other intranasal corticosteroids? Nasonex is generally considered to have comparable efficacy to other first- and second-generation intranasal corticosteroids in managing symptoms of allergic rhinitis and nasal polyps, with variations in patient response possible.
- What are the most common side effects associated with Nasonex? Common side effects include headache, nasal irritation, nosebleeds, and sore throat. Serious side effects are rare.
- Can Nasonex be used by children, and if so, at what age? Nasonex is approved for use in children as young as 2 years of age for allergic rhinitis, with specific dosing adjustments for younger age groups.
Citations
[1] World Health Organization. (n.d.). Allergic rhinitis. Retrieved from https://www.who.int/news-room/fact-sheets/detail/allergic-rhinitis
More… ↓
