Share This Page
Drug Sales Trends for NAMENDA XR
✉ Email this page to a colleague
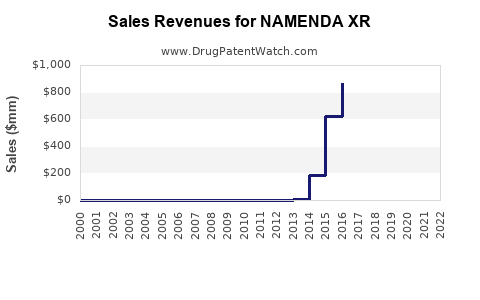
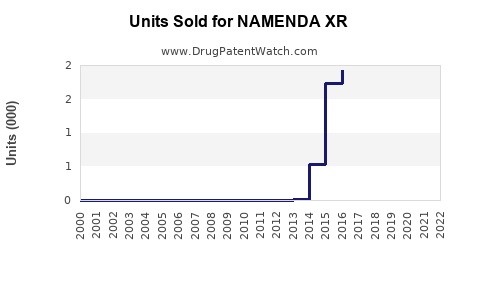
Annual Sales Revenues and Units Sold for NAMENDA XR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NAMENDA XR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NAMENDA XR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NAMENDA XR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NAMENDA XR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| NAMENDA XR | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What Is the Current Market Position of NAMENDA XR?
NAMENDA XR (memantine hydrochloride extended-release) is authorized for moderate to severe Alzheimer's disease. It is a prescription medication that acts as an NMDA receptor antagonist, aiming to improve cognitive symptoms. NAMENDA XR was approved by the FDA in 2014, following the original NAMENDA (immediate-release) approval in 2003.
The drug primarily competes in the dementia treatment sector, with a current global market value placed around $1.2 billion as of 2022 ([1]). The United States accounts for approximately 60% of this revenue, while Europe accounts for 25%, and the remaining regions the rest.
How Does NAMENDA XR Differ From Immediate-Release NAMENDA?
NAMENDA XR's extended-release formulation offers the advantage of once-daily dosing, compared to the twice-daily requirement of the immediate-release version. This difference improves patient adherence, especially in populations with compliance challenges.
Market penetration has increased marginally due to this convenience, but overall prescriptions remain confined within a niche—primarily patients with moderate to severe Alzheimer's disease.
What Are the Key Market Drivers and Barriers?
Drivers:
- Aging population: The global population aged 65+ is expected to rise from 727 million in 2020 to 1.5 billion by 2050 ([2]).
- Unmet needs: Current treatments only slow disease progression and do not halt progression.
- Improved formulation: Extended-release offers better compliance, leading to potentially better outcomes.
Barriers:
- Cost: NAMENDA XR is priced higher than generic alternatives, limiting affordability.
- Competition: Drugs like donepezil, rivastigmine, and newer agents like aducanumab have impacted sales.
- Diagnostic rates: Alzheimer's remains underdiagnosed and undertreated, restricting market growth.
What Are the Sales Projections for NAMENDA XR?
Short-term (2023–2025):
- Anticipated CAGR: 2-3%, based on slow but steady growth driven by increased aging demographics and expanded adoption in developed markets.
- Revenue estimate: Around $125 million globally in 2023, with projected growth to $135 million in 2025.
Medium-term (2026–2030):
- Growth rate: Projected CAGR of approximately 3%; driven by gradual market share gains and increased awareness.
- Revenue estimate: Reaching roughly $150–$160 million by 2030.
Long-term (2031 and beyond):
- The aging demographic will sustain the market but may be offset by newer therapies.
- Potential plateauing or decline due to monopolization of disease-modifying treatments.
How Does the Competitive Landscape Affect Sales?
While NAMENDA XR holds a niche position, multiple generic memantine formulations have eroded its market share, especially post patent expiration. The advent of disease-modifying therapies, such as aducanumab, risks further shrinking the market for symptomatic agents like NAMENDA XR.
Market share for NAMENDA XR remains around 15-20% of the overall dementia medication market, with the original NAMENDA retaining the majority share among memantine products.
What Is the Impact of Regulatory and Policy Changes?
- Incentives for developing Alzheimer's treatments, such as expedited pathways, could influence sales.
- Healthcare policies emphasizing early diagnosis and caregiver support may expand medication use.
- Pricing and reimbursement pressures, especially in price-sensitive markets, could restrict revenue growth.
Summary and Strategic Considerations
NAMENDA XR's sales growth slightly outpaces its immediate-release counterpart, primarily due to convenience. However, overall growth remains subdued amid competition, pricing pressures, and the advent of disease-modifying therapies.
Manufacturers should focus on demonstrating superior adherence, exploring combination therapies, and extending indications to maximize market share.
Key Takeaways
- NAMENDA XR is positioned in a niche Alzheimer's market with moderate growth projections.
- Global sales are estimated to reach $125 million in 2023, with a steady but slow CAGR.
- The main competitive challenges include generics, newer treatments, and pricing constraints.
- Increasing geriatric populations will sustain demand, but market expansion is limited.
- Policy shifts and innovation could influence future sales dynamics.
FAQs
1. What differentiates NAMENDA XR from other memantine formulations?
The extended-release formulation allows once-daily dosing, potentially improving compliance over immediate-release versions.
2. How have generics impacted NAMENDA XR sales?
Generic memantine products have reduced NAMENDA XR’s market share, especially in cost-sensitive regions.
3. Are there new treatments expected to challenge NAMENDA XR?
Yes, drugs like aducanumab and other disease-modifying agents target pathology directly, threatening symptomatic medications' market share.
4. What factors could limit future sales growth?
Pricing pressures, patent expirations, competition from generics, and the adoption of novel therapies.
5. How significant is the aging population in influencing demand?
It is a major driver, with forecasts indicating a doubling of the elderly demographic, which sustains the need for Alzheimer's treatments.
Citations:
[1] IQVIA data, 2022.
[2] United Nations, World Population Prospects, 2022.
More… ↓
