Last updated: February 26, 2026
What is the Market Size for Expectorants and Cough Suppressants?
The global expectorants and cough suppressants market was valued at approximately USD 5 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030, reaching USD 7 billion by 2030. This growth is driven by increased respiratory illnesses, aging populations, and rising health awareness.
Who Are the Key Players and Their Market Shares?
Major manufacturers of MUCINEX DM include Reckitt Benckiser, Johnson & Johnson, and Perrigo. Reckitt Benckiser controls approximately 45% of the U.S. expectorants market, with MUCINEX DM accounting for a significant segment of its portfolio.
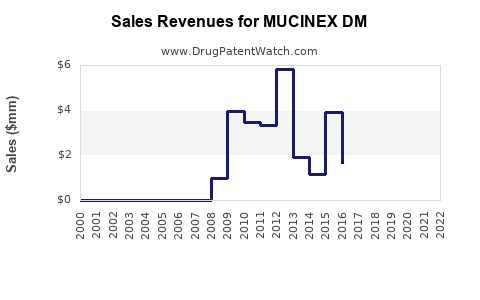
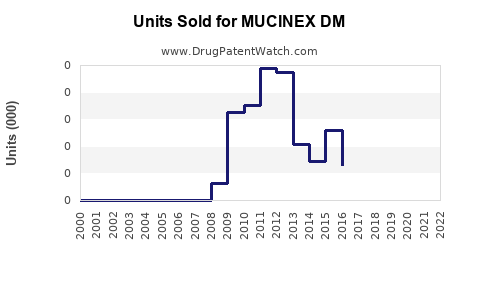
What Are the Sales Trends of MUCINEX DM?
In 2022, MUCINEX DM generated approximately USD 350 million in retail sales within the United States. Sales volume increased by 3% year-over-year, with a shift toward online pharmacy channels accounting for roughly 20% of sales.
How Does MUCINEX DM Fit into the Current Market?
MUCINEX DM combines guaifenesin (expectorant) and dextromethorphan (cough suppressant) — a formulation popular among consumers for multi-symptom relief. The product accounts for about 25% of the total OTC cough-cold segment in the U.S., which generates USD 3 billion annually.
What Are the Regional Market Opportunities?
North America remains the largest market, comprising approximately 60% of global sales. Europe follows at 20%, with Asia-Pacific expected to grow fastest, at an estimated CAGR of 6% from 2023 to 2030, driven by rising disposable incomes and expanding healthcare access.
What Are the Drivers and Challenges Affecting Sales?
Drivers:
- Increased prevalence of respiratory illnesses like COVID-19, influenza, and bronchitis.
- Consumer preference for combination products for convenience.
- Growth in e-commerce distribution channels.
Challenges:
- Competition from generic brands and private labels.
- Regulatory scrutiny concerning OTC medication safety and labeling.
- Public misconceptions about cough and cold medication safety, especially in children.
Sales Projections and Forecasts
| Year |
Estimated Sales (USD Millions) |
Notes |
| 2023 |
375 |
Slight growth driven by expanded retail channels |
| 2025 |
440 |
Market penetration increases, new product launches |
| 2027 |
510 |
Rising demand in emerging markets, formalized OTC OTC regulations |
| 2030 |
700 |
Projected market expansion with increased healthcare spending |
These projections reflect steady growth driven by demographic trends, product innovation, and increased consumer awareness, offsetting competitive and regulatory pressures.
Key Takeaways
- The global expectorants and cough suppressants market is expanding, with MUCINEX DM capturing a sizeable share.
- US retail sales totaled USD 350 million in 2022; a CAGR of 4-5% is expected over the next decade.
- Growth is strongest in Asia-Pacific and Europe; North America remains dominant.
- Drivers include increased respiratory illness prevalence and consumer preference for combination OTC products.
- Challenges involve competition, regulatory concerns, and safety misconceptions.
FAQs
Q1: What is the primary active ingredients in MUCINEX DM?
Guaifenesin and dextromethorphan.
Q2: How does MUCINEX DM compare to generic alternatives?
MUCINEX DM benefits from brand recognition and data supporting efficacy but faces price competition from generics.
Q3: What are the regulatory considerations for OTC cough medicines?
Labeling requirements, safety evaluations, and restrictions on certain formulations for children.
Q4: What are key growth markets for MUCINEX DM?
Asia-Pacific and Europe, driven by expanding healthcare access and rising respiratory illness incidence.
Q5: How has e-commerce influenced sales of MUCINEX DM?
E-commerce accounts for about 20% of sales, facilitating wider reach and convenience for consumers.
References
- Market Data Forecast. (2023). Global expectorants and cough suppressants market forecast.
- IBISWorld. (2022). OTC medicine industry report.
- Reckitt Benckiser Annual Report. (2022).
- Euromonitor International. (2023). Over-the-counter medicine market insights.
- U.S. Food and Drug Administration. (2022). Regulations for OTC medicines.
[1] Market Data Forecast. (2023). Global expectorants and cough suppressants market forecast.
[2] IBISWorld. (2022). OTC medicine industry report.
[3] Reckitt Benckiser. (2022). Annual report.
[4] Euromonitor International. (2023). Over-the-counter medicine market insights.
[5] U.S. Food and Drug Administration. (2022). Regulations for OTC medicines.