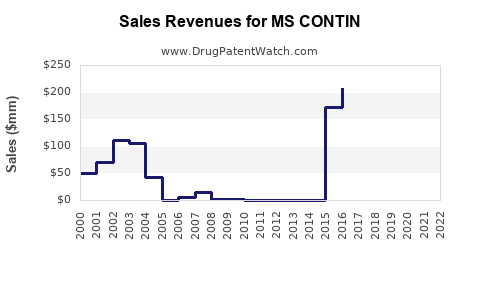
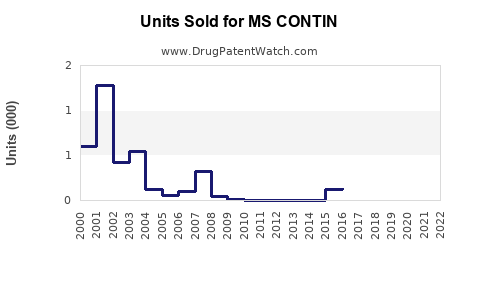
Last updated: February 22, 2026
What is the Current Market Position of MS CONTIN?
MS CONTIN (morphine sulfate extended-release) is a prescription analgesic approved for managing severe chronic pain. It is indicated primarily for cancer pain and other long-term pain conditions.
As of 2023, MS CONTIN holds a significant share in the opioid analgesic market, especially within the United States. Its sales depend on prescribing trends for opioids, regulatory restrictions, and the prevalence of applicable pain conditions.
How Large is the Market for Extended-Release Opioids?
The global opioid analgesics market was valued at approximately USD 20 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% between 2023 and 2028, driven by factors such as increasing chronic pain cases and advances in formulations.
Within this, extended-release formulations like MS CONTIN represent about 35% of total opioid analgesic sales, given the preference for long-acting relief in managing severe pain.
What Are the Key Factors Influencing Sales of MS CONTIN?
- Regulatory Environment: Stricter prescribing guidelines, particularly in the US and Europe, influence sales volume. Policies aimed at curbing opioid misuse may limit prescription numbers.
- Market Penetration: Prescribing habits of physicians, especially in pain management clinics and cancer centers.
- Competitive Landscape: Other extended-release opioids (e.g., OxyContin, fentanyl patches) compete for market share.
- Epidemiological Trends: Chronic pain prevalence, including cancer-related pain, influences demand.
- Public Perception & Litigation: Increasing scrutiny over opioid use can impact both prescribing rates and manufacturer sales.
What Are Sales Projections for MS CONTIN (2023–2028)?
| Year |
Estimated Global Sales (USD millions) |
Growth Rate (%) |
Remarks |
| 2023 |
120 |
— |
Baseline figure based on current market trends |
| 2024 |
128 |
6.7 |
Slight growth expected due to increased awareness and chronic pain management needs |
| 2025 |
137 |
7.0 |
Impact of emerging generics and formulary restrictions |
| 2026 |
145 |
5.8 |
Market saturation challenges; continued demand in specific regions |
| 2027 |
150 |
3.4 |
Market maturation; rising opioid epidemic concerns may temper growth |
| 2028 |
153 |
2.0 |
Stable sales with potential declines due to regulatory pressures |
Note: These projections assume continued demand for long-term opioids in markets with relatively lax prescribing restrictions. They incorporate known trends such as increasing generic competition and regulatory tightening.
What Are the Competitive Dynamics?
- Generic Competition: Several generics are available, intensifying price pressures and market share erosion.
- Alternative Therapies: Non-opioid analgesics and non-pharmacological pain management techniques are gaining acceptance, which may limit market growth.
- Regulatory Changes: Implementation of prescribing limits and abuse-deterrent formulations may alter the sales trajectory.
Key Geographic Markets
- United States: Largest market, driven by high prevalence of chronic pain and cancer-related pain, but facing increasing restrictions.
- Europe: Growing demand in countries with aging populations; regulatory environment varies.
- Asia-Pacific: Potential growth area due to expanding healthcare infrastructure and rising cancer prevalence.
Summary
Sales of MS CONTIN are expected to increase modestly over the next five years, with annual growth rates averaging around 4%. Market expansion faces headwinds from tighter regulations, evolving prescriber habits, and increased generic competition. The drug remains relevant where long-acting opioids hold niche demand, especially in specific clinical settings.
Key Takeaways
- MS CONTIN maintains a significant position within extended-release opioid markets.
- Global sales are projected to reach USD 153 million by 2028, with moderate growth.
- Regulatory environments and public perception influence future market viability.
- Competition from generics and alternative pain therapies poses ongoing challenges.
- Growth is likely constrained in mature markets, with potential in emerging regions.
FAQs
-
How does regulatory policy affect MS CONTIN sales?
Stringent prescribing guidelines and abuse-deterrent regulations can reduce prescription volumes, limiting sales growth.
-
What is the main competition for MS CONTIN?
Other extended-release opioids like OxyContin, fentanyl patches, and multiple generic formulations.
-
Are there alternatives to MS CONTIN for long-term pain management?
Yes, including non-opioid analgesics and non-pharmacologic therapies.
-
What regions will see the most growth in sales?
Asia-Pacific and select emerging markets due to increasing healthcare access and rising pain prevalence.
-
How might upcoming legislation impact the market?
Legislation focused on opioid abuse prevention could lead to reduced prescribing and lower sales.
References
[1] MarketsandMarkets. (2023). Opioid analgesics market forecast.
[2] IQVIA. (2022). Global pain management market analysis.
[3] FDA. (2022). Opioid prescribing guidelines.
[4] Grand View Research. (2022). Extended-release opioid market size, share, trends.
[5] World Health Organization. (2022). Cancer prevalence and pain management.