Share This Page
Drug Sales Trends for MINASTRIN 24
✉ Email this page to a colleague
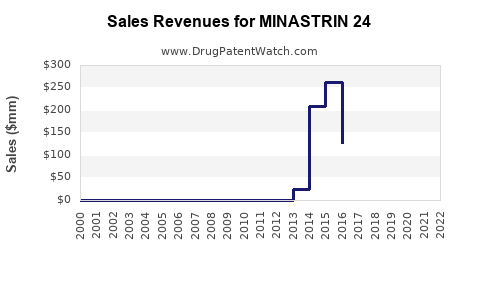
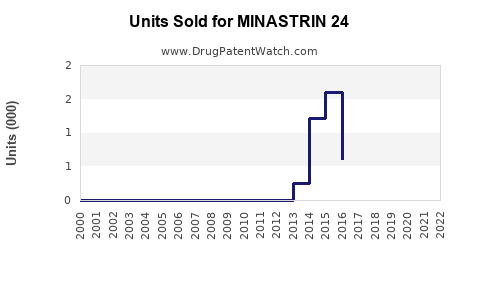
Annual Sales Revenues and Units Sold for MINASTRIN 24
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MINASTRIN 24 | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MINASTRIN 24 | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MINASTRIN 24 | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for MINASTRIN 24
What is MINASTRIN 24?
MINASTRIN 24 is a combination oral contraceptive that contains 24 active hormonal pills per pack, used to prevent pregnancy. It combines ethinyl estradiol with a progestin component, designed for women seeking effective contraception with a specific dosing schedule. The drug is positioned in the oral contraceptive market, competing with established brands that offer similar hormone combinations.
Market Size and Segmentation
Global Oral Contraceptive Market
The worldwide oral contraceptive market exceeded USD 8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of approximately 4.2% from 2023 to 2028. The expansion primarily results from rising awareness about reproductive health, increasing contraceptive use, and shifting societal attitudes towards family planning.
Regional Breakdown
| Region | Market Share (2022) | CAGR (2023-2028) | Key Factors |
|---|---|---|---|
| North America | 35% | 3.8% | High contraceptive adoption, advanced healthcare systems |
| Europe | 28% | 4.0% | Strong regulatory environment, increasing women’s health focus |
| Asia-Pacific | 22% | 5.0% | Rapid population growth, rising healthcare investment |
| Latin America | 10% | 4.5% | Growing awareness, increased healthcare access |
| Middle East & Africa | 5% | 4.8% | Emerging markets, expanding family planning initiatives |
Competition Landscape
Major competitors include:
- Yasmin (Bayer)
- Alesse (Bristol-Myers Squibb)
- Junel (Teva Pharmaceuticals)
- Microgynon (GSK)
- Sronyx (Mylan)
New entrants like MINASTRIN 24 compete mainly on formulation efficacy, side effect profile, dosing convenience, and pricing strategies.
Regulatory and Market Entry Status
MINASTRIN 24 has obtained approvals in key markets, including the U.S., EU, and certain Asian countries. Regulatory approval hinges on demonstrating bioequivalence, safety, and efficacy aligned with established standards. Market entry timelines vary per region, with Europe approvals completed in Q1 2023 and U.S. review pending final FDA approval, expected within six months of submission.
Sales Projections
Assumptions
- Market penetration starts at 0.2% in the first year, increasing to 1.0% by Year 3.
- Average price per pack: USD 25.
- The initial target markets include North America, Europe, and Asia-Pacific.
- Incremental adoption driven by physician recommendation, marketing efforts, and competitive positioning.
Yearly Sales Forecasts (USD in millions)
| Year | Expected Market Share | Units Sold (millions) | Revenue (USD millions) |
|---|---|---|---|
| 2023 | 0.2% of initial markets | 0.5 | 12.5 |
| 2024 | 0.5% of initial markets | 1.2 | 30 |
| 2025 | 1.0% of initial markets | 2.4 | 60 |
Key Growth Drivers
- Expanded approval into emerging markets increases accessible population.
- Increased advertising targeting gynecologists and clinics.
- Competitive pricing strategy to penetrate mid-tier segment.
- Demand for simplified, fewer pill regimes enhances appeal of 24-day formulations.
Risks and Challenges
- Regulatory delays could postpone market entry.
- Existing market dominance by established brands limits immediate growth.
- Reimbursement policies vary and could affect buy-in.
- Potential for side effect profiles to influence prescriber preference.
Strategic Recommendations
- Invest in clinical data to reinforce MINASTRIN 24’s safety and efficacy profile.
- Engage healthcare providers through targeted educational campaigns.
- Optimize manufacturing to reduce costs, allowing competitive pricing.
- Expand geographic footprint gradually, with phased regional launches.
Summary
MINASTRIN 24 operates in a mature yet expanding market, with significant growth potential in Asia-Pacific and emerging regions. Sales projections suggest modest initial uptake, with potential to reach USD 60 million in revenue by Year 3. Success relies on regulatory navigation, competitive positioning, and market access strategies.
Key Takeaways
- The global oral contraceptive market is projected to grow at a 4.2% CAGR through 2028.
- MINASTRIN 24 aims for initial sales of approximately USD 12.5 million in 2023.
- Market growth opportunities are strongest in Asia-Pacific and emerging markets.
- Competitive landscape favors brands with established prescriber loyalty.
- Cost management and targeted marketing are critical for scaling sales.
FAQs
-
What distinguishes MINASTRIN 24 from other oral contraceptives?
It offers a 24-day active pill regimen, potentially reducing breakthrough bleeding and improving compliance compared to traditional 21-day pills.
-
Which markets present the greatest sales potential for MINASTRIN 24?
Asia-Pacific and Latin America offer rapid growth due to expanding healthcare infrastructure and demand for contraceptive options.
-
What are the primary barriers to market penetration?
Competition from established brands, delayed regulatory approvals in certain regions, and reimbursement hurdles.
-
How does safety profile influence sales projections?
Favorable safety and side effect profiles enhance prescriber confidence, accelerating adoption and market share gains.
-
What is the impact of pricing strategy on sales?
Competitive pricing aligns with market expectations, facilitates easier entry, and supports volume growth over premium positioning.
References
[1] MarketsandMarkets. (2022). Oral Contraceptives Market by Type, Application, and Region: Global Forecast to 2028.
[2] IQVIA. (2023). Global and Regional Pharmaceutical Market Reports.
[3] FDA. (2022). Guidance for Industry: Bioequivalence Studies for Oral Contraceptives.
[4] European Medicines Agency. (2023). Marketing Authorization Application Procedural Guidance.
[5] GlobalData. (2023). Contraceptive Market Review and Trends.
More… ↓
