Share This Page
Drug Sales Trends for MEVACOR
✉ Email this page to a colleague
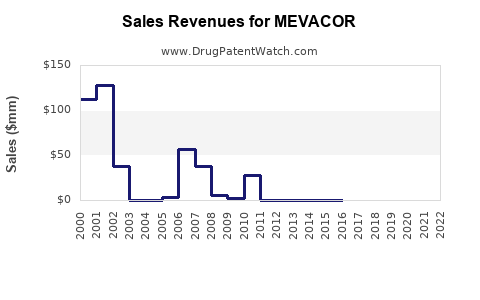
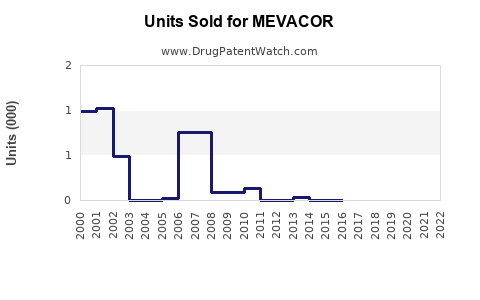
Annual Sales Revenues and Units Sold for MEVACOR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MEVACOR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MEVACOR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MEVACOR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MEVACOR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for MEVACOR (Lovastatin)
What is the Current Market Position of MEVACOR?
MEVACOR (lovastatin) is a statin prescribed to lower low-density lipoprotein (LDL) cholesterol and reduce cardiovascular risk. It was introduced in 1987 by Merck & Co. and became among the initial statins available. The drug's patent expired in many markets by 2009, leading to generic availability. Consequently, MEVACOR's market share has declined due to competition from branded and generic alternatives.
Market Size and Dynamics
The global cholesterol-lowering drug market was valued at approximately USD 22 billion in 2022. Statins account for roughly 70% of this market but have seen growth slowing. Merck's branded position eroded post-patent expiry, with generic lovastatin capturing a significant portion. The focus shifts to long-term usage, differentiated formulations, and regional market penetration.
Key factors influencing market size:
- Prevalence of hypercholesterolemia: Estimated at 30% of adults globally.
- Cardiovascular disease (CVD) burden: Accounted for 32% of all deaths in 2019.
- Drug penetration: Generic statins dominate prescription patterns in developed markets, with incremental growth in developing markets due to increasing CVD awareness.
Competitive Landscape
The cholesterol medication market consists of several key players:
| Company | Product(s) | Market Share (2022) | Notes |
|---|---|---|---|
| Pfizer | Lipitor (atorvastatin) | 35% | Dominates in high-potency statins |
| Novartis | Rosuvastatin (Crestor) | 25% | Higher potency, preferred in some indications |
| Merck | Lovastatin (MEVACOR) | Declining | Generic availability since 2009 |
| Mylan (now part of Viatris) | Lovastatin (generic) | Growing | Key supplier in generics |
| Others (Sun, Teva) | Various statins | 15% combined | Regional presence |
Sales Projections
Sales of MEVACOR peaked in the late 1980s and early 1990s. Post-patent expiration, annual sales declined sharply:
| Year | Estimated Global Sales (USD millions) | Notes |
|---|---|---|
| 2009 | 150 | Post-patent expiry peak for generic lovastatin |
| 2010 | 80 | Decline as generics gained market share |
| 2015 | 10 | Marginal contribution remaining |
| 2020 | 5 | Almost negligible, replaced by generics |
| 2022 | 3-5 | Minor niche use in specific regions |
Future sales are unlikely to recover absent new formulations or indication expansions. Adoption of other statins like atorvastatin and rosuvastatin captured most of the market share.
Regional Market Trends
- North America: Mature market with high generic penetration. Annual sales of generics almost exclusively.
- Europe: Similar to North America, with some regions still utilizing MEVACOR for specific indications.
- Asia-Pacific: Growing market for all statins due to increasing CVD prevalence and improving healthcare access. Small niche use of MEVACOR persists, but most prescriptions favor newer agents.
- Emerging Markets: High potential but limited immediate use; cost sensitivity favors generics over branded MEVACOR.
Regulatory and Policy Factors
- Patents for lovastatin expired in 2009, enabling widespread generic manufacture.
- Regulatory agencies (FDA, EMA) promote bioequivalence standards, facilitating generic uptake.
- Some regional restrictions apply for initial indications, but overall, MEVACOR's role diminishes significantly.
Key Market Drivers and Barriers
| Drivers | Barriers |
|---|---|
| Rising prevalence of CVD | Availability of more potent and newer statins |
| Cost advantages of generics | Limited indications for MEVACOR compared to newer agents |
| Increased awareness of cholesterol management | Reduced physician prescription due to efficacy of alternatives |
| Established safety profile (original formulation) | Diminished marketing and promotional activities |
Opportunities for MEVACOR
Limited unless repositioned for niche roles or formulation innovations. The primary value lies in generic supply, low-cost formulations, and potential use in specific populations where newer statins are contraindicated.
Summary
MEVACOR's market presence is relegated to niche segments and regional remnants. Its sales continue to decrease and are unlikely to rebound without significant repositioning or innovation. The broader cholesterol treatment market is dominated by atorvastatin and rosuvastatin, with generic versions capturing nearly all of the volume.
Key Takeaways
- MEVACOR's peak sales occurred pre-2009; current sales are marginal.
- The market shifted rapidly post-patent expiry to generic statins, diminishing MEVACOR's sales.
- The global cholesterol market is highly competitive, with high patent and generic saturation.
- Future growth for MEVACOR is limited to niche applications; no significant pipeline or reformulation appears imminent.
- Companies focusing on innovative lipid-lowering therapies hold more growth potential than traditional statins like MEVACOR.
FAQs
1. Will MEVACOR regain market share through new formulations?
No. The primary advantage of MEVACOR, the original drug, is diminished by generics. Reformulation or new indications would be necessary but are unlikely given the current market landscape.
2. What are the main competitors replacing MEVACOR?
Atorvastatin (Lipitor) and rosuvastatin (Crestor) lead the market, offering higher potency, fewer dosages, and expanded indications.
3. How does the global prevalence of hypercholesterolemia impact MEVACOR sales?
While prevalence increases demand for cholesterol-lowering drugs, MEVACOR's market share shrinks due to the rise of more potent and newer statins, especially in developed markets.
4. Are there regional markets where MEVACOR still has significant sales?
Small residual markets exist in some regions with limited access to newer medications or preference for older formulations; however, these are declining.
5. Are there upcoming patent protections or exclusivities for MEVACOR?
No. The original patent expired in 2009, and there are no new patent protections. The drug is available as a generic worldwide.
References
[1] IMS Health (2022). Global Prescription Drug Market Data.
[2] IQVIA (2022). Market Data Archives.
[3] U.S. Food and Drug Administration (2009). Lovastatin Patent Expiry and Generic Entry.
[4] WHO (2019). Cardiovascular Disease Statistics.
[5] MarketsandMarkets (2022). Cholesterol-Lowering Drugs Market Report.
More… ↓
