Share This Page
Drug Sales Trends for METRONIDAZOLE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for METRONIDAZOLE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
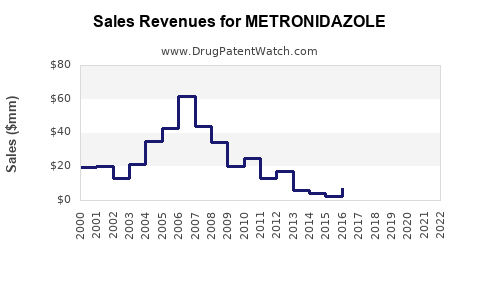
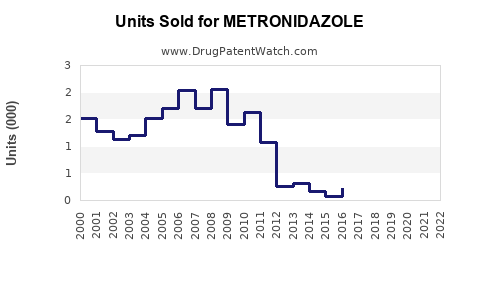
Annual Sales Revenues and Units Sold for METRONIDAZOLE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METRONIDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METRONIDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METRONIDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METRONIDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| METRONIDAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Metronidazole: Market Landscape and Sales Forecast
This analysis examines the global market for metronidazole, focusing on its patent landscape, current sales performance, and future projections. Metronidazole is a widely used antibiotic and antiprotozoal medication with a long history of market presence. Its established efficacy, broad spectrum of activity, and affordability contribute to its sustained demand, particularly in treating anaerobic bacterial and protozoal infections.
What is the Current Global Market Size for Metronidazole?
The global metronidazole market size was valued at approximately $250 million to $300 million in 2023. This valuation is based on reported sales figures and market research data from pharmaceutical industry analysts. The market demonstrates consistent demand driven by the prevalence of infections treated by metronidazole, such as bacterial vaginosis, trichomoniasis, amebiasis, and anaerobic infections in surgical settings.
Key factors influencing market size include:
- Therapeutic indications: Metronidazole is prescribed for a range of gastrointestinal, gynecological, dermatological, and surgical site infections.
- Generic availability: Metronidazole is largely a generic drug, with numerous manufacturers producing it. This competition keeps prices low but also ensures widespread accessibility.
- Geographic distribution: Demand is significant in both developed and developing economies, with a substantial portion of sales attributed to regions with higher incidences of parasitic and anaerobic infections.
What is the Patent Landscape for Metronidazole?
Metronidazole was first patented in the 1950s. As a mature drug, its original composition of matter patents have long expired. Consequently, there are no active patents on the core metronidazole molecule that would prevent generic manufacturing and sales.
Current patent activity, if any, would primarily focus on:
- New formulations: Improved delivery systems, extended-release versions, or combination therapies incorporating metronidazole.
- Novel manufacturing processes: More efficient or cost-effective synthesis methods.
- New therapeutic uses: Discovery of metronidazole's efficacy in treating conditions beyond its current approved indications.
However, these secondary patents have not significantly altered the competitive landscape of metronidazole due to the drug's established generic status and the availability of numerous alternative treatments for many of its core indications. The primary barrier to entry for new market participants is not patent protection but rather manufacturing capabilities and regulatory compliance.
What are the Key Therapeutic Indications and Their Market Impact?
Metronidazole's market demand is directly linked to the prevalence and treatment guidelines for specific infections. The most significant indications include:
- Bacterial Vaginosis (BV): This is a leading indication, particularly in women's health. The high recurrence rate of BV contributes to ongoing prescription volumes.
- Trichomoniasis: A common sexually transmitted infection, requiring metronidazole treatment. Public health initiatives and screening programs can influence demand.
- Amebiasis: Endemic in many developing regions, amebiasis remains a significant driver of metronidazole sales, especially in oral and injectable forms for severe cases.
- Anaerobic Infections: This broad category includes infections in the abdomen, pelvis, skin, soft tissues, and bones. Metronidazole is a first-line or adjunctive therapy for many of these serious infections, particularly in hospital settings.
- Clostridioides difficile Infection (CDI): While vancomycin is often preferred for severe CDI, metronidazole can be used for milder cases or in specific patient populations.
- Prophylaxis in Surgery: Used to prevent anaerobic infections following certain types of surgery, such as colorectal and gynecological procedures.
The relative impact of each indication can vary by region. For instance, amebiasis has a larger market share in parts of Africa, Asia, and Latin America, while BV and CDI contribute more significantly to demand in North America and Europe.
Who are the Major Manufacturers and Competitors in the Metronidazole Market?
The metronidazole market is highly fragmented due to its generic nature. Major manufacturers include:
- Bayer AG: While an originator of metronidazole, their current market presence is largely through branded generics or supply agreements.
- Generic Pharmaceutical Companies: A vast number of companies globally produce generic metronidazole. Prominent players in the broader antibiotic and generic drug space that likely have metronidazole in their portfolios include:
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc. (formed from the merger of Mylan and Pfizer's Upjohn)
- Sanofi S.A.
- Sun Pharmaceutical Industries Ltd.
- Apotex Inc.
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (prior to merger with Upjohn)
These companies compete primarily on price, manufacturing scale, and distribution networks. The availability of multiple suppliers ensures a competitive pricing environment.
What are the Sales Projections and Market Growth Rate for Metronidazole?
The global metronidazole market is projected to experience modest growth, with a compound annual growth rate (CAGR) of approximately 2% to 3% over the next five to seven years. This stable, albeit low, growth is driven by several factors:
- Sustained Demand: The continued prevalence of anaerobic and protozoal infections ensures a baseline demand for metronidazole.
- Affordability: Its low cost makes it a critical treatment option in resource-limited settings, underpinning consistent sales volume.
- Established Treatment Protocols: Metronidazole remains a cornerstone therapy in many clinical guidelines.
However, the market faces limitations that temper higher growth:
- Emergence of Newer Antibiotics: For some indications, newer, more potent, or narrower-spectrum antibiotics are gaining preference, potentially displacing metronidazole.
- Antibiotic Resistance: While not as pronounced as with some other antibiotics, concerns about resistance could influence prescribing patterns.
- Competition from Alternatives: For certain indications like BV, alternative treatments exist and may be preferred by some prescribers or patients.
- Pricing Pressures: Intense competition among generic manufacturers leads to significant pricing pressure, limiting revenue growth even with stable volumes.
Projected Market Size:
| Year | Estimated Market Value (USD Billions) |
|---|---|
| 2024 | $255 - $310 |
| 2026 | $265 - $325 |
| 2028 | $275 - $340 |
| 2030 | $285 - $355 |
These figures represent anticipated revenue based on current market trends and do not account for unforeseen disruptions such as major outbreaks of metronidazole-responsive infections or significant shifts in global healthcare policy.
What are the Regulatory and Clinical Considerations Affecting the Metronidazole Market?
Regulatory approvals and clinical guidelines play a critical role in the metronidazole market.
- FDA (U.S. Food and Drug Administration) & EMA (European Medicines Agency): Metronidazole is approved for various indications by major regulatory bodies. Post-market surveillance continues to monitor its safety and efficacy.
- Clinical Guidelines: Recommendations from organizations such as the Infectious Diseases Society of America (IDSA) or the World Health Organization (WHO) significantly influence prescribing practices. Changes in these guidelines, for example, regarding first-line treatment for specific infections, can impact metronidazole's market share.
- Adverse Event Reporting: As with all medications, adverse event reporting systems monitor for and evaluate potential safety concerns. Significant findings could lead to label changes or restricted use.
- Antibiotic Stewardship Programs: These programs aim to optimize antibiotic use. While metronidazole is generally considered a safe and effective option, stewardship principles encourage its appropriate use, avoiding unnecessary prescriptions and promoting timely de-escalation where appropriate.
Key Takeaways
- The global metronidazole market is a mature, stable segment valued at approximately $250-300 million annually.
- Original composition of matter patents have expired, making metronidazole a widely available generic drug with no significant patent-based barriers to entry.
- Demand is driven by established indications like bacterial vaginosis, trichomoniasis, amebiasis, and anaerobic infections.
- The market is highly competitive, with numerous generic manufacturers focusing on price and volume.
- Future growth is projected at a modest CAGR of 2-3%, primarily supported by consistent demand in developing regions and its affordability.
- Regulatory approvals and evolving clinical guidelines remain key influencers of prescribing patterns and market dynamics.
Frequently Asked Questions
Is metronidazole still considered a first-line treatment for bacterial vaginosis?
Yes, metronidazole, particularly in oral and vaginal formulations, remains a widely recommended first-line treatment for bacterial vaginosis by major health organizations.
What is the impact of antibiotic resistance on metronidazole sales?
While antibiotic resistance is a concern for all antimicrobial agents, metronidazole has historically maintained good efficacy against its primary targets. However, increasing resistance could lead to reduced efficacy in certain geographic areas or for specific patient populations, potentially impacting its long-term market share.
Are there any significant new therapeutic uses for metronidazole currently under development?
While research into novel applications of existing drugs is ongoing, there are no widely publicized, late-stage clinical trials indicating significant new therapeutic uses for metronidazole that are expected to dramatically alter its market trajectory in the near future.
How does metronidazole pricing compare to newer antibiotics used for similar infections?
Metronidazole is a generic drug and is significantly less expensive than most newer, branded antibiotics, making it a critical treatment option, especially in low- and middle-income countries.
What are the primary geographic regions driving metronidazole sales?
Sales are driven by both developed and developing economies. However, regions with higher prevalence of parasitic infections like amebiasis, such as parts of Asia, Africa, and Latin America, represent substantial markets alongside significant demand for indications like bacterial vaginosis in North America and Europe.
Citations
[1] Global Antibiotics Market Report. (2023). Mordor Intelligence. [2] Metronidazole Market Analysis. (2023). Grand View Research. [3] Antibiotic Market Share. (2023). Statista. [4] Infectious Diseases Society of America (IDSA) Guidelines for the treatment of bacterial vaginosis and trichomoniasis. (Accessed 2024). [5] World Health Organization (WHO) Model List of Essential Medicines. (Accessed 2024).
More… ↓
