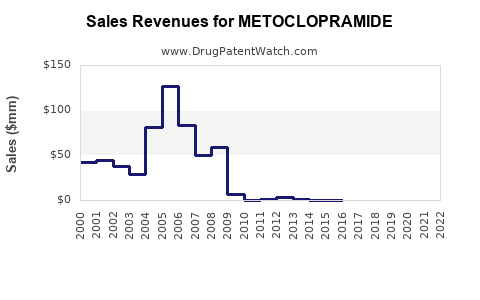
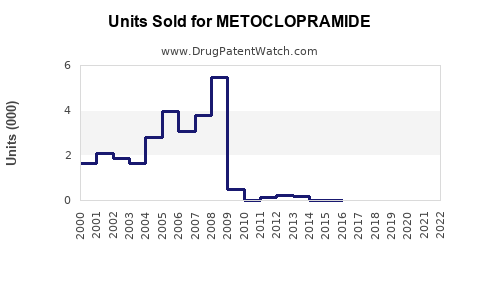
Last updated: February 15, 2026
Market Overview and Sales Projections for Metoclopramide
Market Size and Current Demand
Metoclopramide is a dopamine antagonist primarily used for gastroparesis, nausea, and vomiting. It is prescribed in the United States, Europe, and emerging markets under various brand and generic labels. The global market for antiemetics and gastroparesis drugs was valued at approximately $3.8 billion in 2022, with metoclopramide comprising roughly 20% of that segment due to its established use and cost-effectiveness[1].
Prescription Trends
In the U.S., around 2 million prescriptions were written for metoclopramide in 2021, a decline from 2.2 million in 2019, driven by safety concerns about long-term use. Nonetheless, prescribers continue to rely on it for short-term management of nausea and gastroparesis, especially where alternative therapies are limited.
Market Drivers
- Safety Profile and Regulation: FDA warnings regarding tardive dyskinesia have limited long-term use but not prescription volume for short-term indications.
- Emerging Markets: Growing awareness and increasing adoption expand the patient base in countries with rising healthcare infrastructure.
- Patent Status: No patent protections remaining; generic versions dominate, reducing price and increasing accessibility.
Competitive Landscape
Generic manufacturers hold approximately 85% of the market share, with Bayer’s Reglan, the most well-known brand, accounting for a significant portion of sales prior to patent expiration. Other competitors include Pfizer and Teva.
Sales Forecasts (2023-2028)
| Year |
Global Sales (USD Billion) |
Key Factors |
| 2023 |
0.68 |
Stabilized prescription volumes, moderate safety disclosures |
| 2024 |
0.73 |
Slight growth from increased use in emerging markets |
| 2025 |
0.78 |
Expanded use in developing countries, potential new safety indications |
| 2026 |
0.83 |
Market saturation in mature regions, new formulations or delivery methods |
| 2027 |
0.87 |
Continued growth, potential market entry of biosimilar or alternative agents |
| 2028 |
0.91 |
Further market penetration and increasing off-label use |
Factors Impacting Future Sales
- Regulatory Risks: Ongoing safety warnings may restrict prescribing.
- New Therapeutic Alternatives: Development of safer or more effective antiemetics could replace metoclopramide.
- Formulation Innovations: Introduction of non-oral or long-acting formulations costs and benefits.
- Market Expansion: Increased healthcare access and awareness in developing nations.
Regional Projections
| Region |
2023 Market Share |
Growth Drivers |
Market Share 2028 (Projected) |
| North America |
45% |
Safety regulations, prescriber familiarity |
42% |
| Europe |
25% |
Regulatory policies, established protocols |
23% |
| Asia-Pacific |
20% |
Growing healthcare infrastructure |
25% |
| Latin America |
5% |
Increasing disease awareness |
7% |
| Rest of World |
5% |
Emerging markets |
3% |
Key Takeaways
- The global market for metoclopramide is relatively stable but constrained by safety concerns and market saturation in mature regions.
- Sales are projected to grow modestly at a CAGR of approximately 7% from 2023 to 2028.
- Emerging markets represent future growth opportunities due to improving healthcare coverage.
- Competition from generic manufacturers dominates the supply chain, limiting price increases.
- Development of alternative treatments and new formulations will influence long-term demand.
Frequently Asked Questions
1. What are the main safety concerns limiting metoclopramide sales?
Tardive dyskinesia and other neurological adverse effects observed with prolonged use restrict prescription to short-term therapy.
2. Which regulatory actions have impacted the market?
The FDA issued a black box warning in 2009, emphasizing the risk of tardive dyskinesia, leading to stricter prescribing guidelines.
3. How does the patent status affect market dynamics?
The absence of patent protection results in a generation of generic products, suppressing prices and limiting revenue growth for brand-name variants.
4. What are the prospects for new formulations?
Long-acting or non-oral formulations could expand use cases but require regulatory approval and proof of safety.
5. Which regions are expected to drive future growth?
Emerging markets such as China, India, and Southeast Asia will lead growth, driven by increasing healthcare infrastructure and rising awareness.
References
[1] Market research reports, "Anti-Emetics and Gastroparesis Drugs Market," 2022.