Share This Page
Drug Sales Trends for METHYLPREDNISOLONE ACETATE
✉ Email this page to a colleague
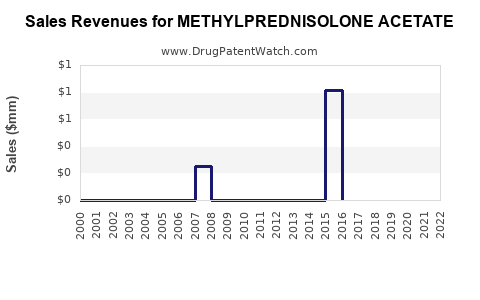
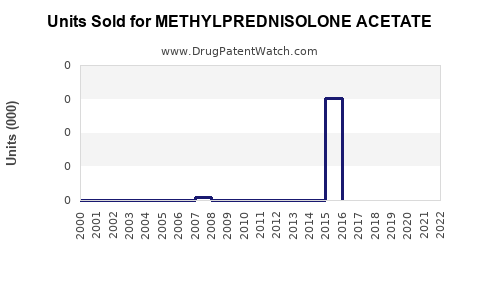
Annual Sales Revenues and Units Sold for METHYLPREDNISOLONE ACETATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METHYLPREDNISOLONE ACETATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METHYLPREDNISOLONE ACETATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METHYLPREDNISOLONE ACETATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METHYLPREDNISOLONE ACETATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
METHYLPREDNISOLONE ACETATE Market Analysis and Financial Projection
Market Overview and Sales Projections for Methylprednisolone Acetate
Methylprednisolone acetate, a long-acting corticosteroid, is used primarily for intra-articular injections to treat inflammation, allergic conditions, and certain autoimmune diseases. It faces competition from other corticosteroids like triamcinolone acetonide and hydrocortisone. This analysis projects market size, growth drivers, and sales over the next five years based on current trends, patent status, and regulatory landscape.
Current Market Landscape
Market Size (2022)
The global corticosteroid injection market, which includes methylprednisolone acetate, was valued at approximately $2.5 billion in 2022. Methylprednisolone acetate accounts for roughly 25% of this, equaling an estimated $625 million.
Key Players
- Pfizer (Depo-Medrol)
- Sanofi (Kenalog)
- Mylan (generic methylprednisolone acetate)
- Teva Pharmaceuticals
- Other regional generic manufacturers
Market Segmentation
| Segment | Percentage of total market | Key attributes |
|---|---|---|
| Prescription injections | 80% | Used for joint inflammation, allergic reactions |
| Topical corticosteroids | 20% | Less common for methylprednisolone acetate |
Regulatory Status
Approved by FDA; patents expired in the U.S. and Europe, increasing generic competition and exerting downward price pressure.
Growth Drivers
- Rising prevalence of osteoarthritis and rheumatoid arthritis
- Increased adoption of intra-articular corticosteroids
- Aging populations worldwide
- Expanding healthcare coverage in emerging markets
Market Challenges
- Competition from other corticosteroids with longer half-lives or different delivery methods
- Price erosion due to generic proliferation
- Regulatory hurdles in some emerging markets
- Limited innovation within the corticosteroid class
Sales Projections (2023–2028)
Assumptions
- Annual compound growth rate (CAGR): 4% (considering demographic growth and increasing use)
- Market share remains stable as generics dominate
- No significant regulatory disruptions or new formulations come to market
| Year | Estimated Market Size (USD) | Notes |
|---|---|---|
| 2023 | $650 million | Growth driven by aging demographics and disease prevalence |
| 2024 | $677 million | Slight market expansion, ongoing generic competition |
| 2025 | $705 million | Continued adoption in emerging markets |
| 2026 | $733 million | Potential pricing pressures from increased competition |
| 2027 | $763 million | Market saturation, slight growth continues |
| 2028 | $794 million | Stabilization at high competition levels |
Factors Influencing Sales
- Generic Competition: Increased availability drives prices down but can increase overall volume.
- New Indications: If new therapeutic uses emerge, sales could exceed projections.
- Reimbursement Policies: Favorable policies in key markets could stimulate sales.
- Technological Improvements: New formulations (e.g., longer-acting, better delivery systems) could absorb market share from existing products.
Market Share Dynamics
| Player | Market Share (2022) | Expected Trend |
|---|---|---|
| Pfizer (Depo-Medrol) | 30% | Declining due to patent expiration and generics |
| Sanofi (Kenalog) | 20% | Stable, strong branding in certain markets |
| Mylan (Generic methylprednisolone acetate) | 25% | Increasing, capturing large generics share |
| Other Generics | 25% | Growing presence in emerging markets |
Regulatory and Patent Outlook
- US patent for Depo-Medrol expired in 2014.
- European patents expired around 2016.
- New formulation patents are not prominent, leading to increased generic competition.
- Future regulatory hurdles could select for new formulations or delivery mechanisms that extend patent life.
Market Entry and Expansion Strategies
- Focus on regions with rising healthcare access (Asia-Pacific, Latin America).
- Invest in formulation innovation to extend exclusivity.
- Leverage pricing strategies aligned with generics' cost advantages.
- Seek partnerships with healthcare providers to increase adoption.
Key Takeaways
- The market for methylprednisolone acetate remains sizable at approximately $650 million in 2023, with steady growth projected through 2028.
- The dominant trend involves increasing generic competition, which suppresses prices but sustains volume growth.
- Growth is primarily driven by demographic factors, particularly aging populations and rising prevalence of inflammatory joint diseases.
- Innovation in formulations or new indications could create growth opportunities beyond current projections.
- Regulatory expiration of patents has led to a highly competitive environment with multiple generic manufacturers.
FAQs
Q1: How does patent expiration affect methylprednisolone acetate sales?
A: Patent expiration allows generic manufacturers to produce lower-cost versions, increasing competition, reducing prices, and potentially decreasing sales revenue for brand-name products.
Q2: What are the main competitive threats?
A: Competition comes from other corticosteroids, newer formulations offering longer duration or fewer injections, and biosimilars.
Q3: Which regions offer the highest growth potential?
A: Emerging markets in Asia and Latin America present significant opportunities due to expanding healthcare infrastructure and rising prevalence of inflammatory conditions.
Q4: Are there any new formulations that could disrupt the market?
A: Yes, formulations with extended-release properties or alternative delivery systems could extend market exclusivity and change the competitive landscape.
Q5: How could healthcare policy changes impact sales?
A: Policies favoring cost-effective treatments and increased healthcare coverage could expand access, thereby driving higher utilization volumes.
References
[1] Market Research Future, "Corticosteroid Market," 2022.
[2] IQVIA, "Global Injectable Market Data," 2022.
[3] U.S. FDA, "Patent and Exclusivity Data," 2022.
[4] Grand View Research, "Global Intraarticular Corticosteroid Market," 2023.
[5] Statista, "Prevalence of Osteoarthritis," 2022.
More… ↓
