Share This Page
Drug Sales Trends for METHYLIN
✉ Email this page to a colleague
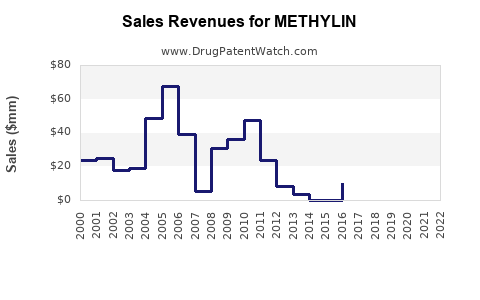
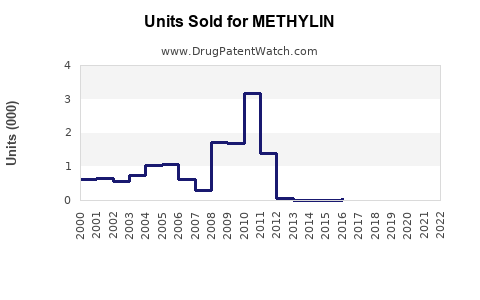
Annual Sales Revenues and Units Sold for METHYLIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METHYLIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METHYLIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METHYLIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| METHYLIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for METHYLIN (Methylphenidate)
What Is Methylphenidate and How Is It Used?
Methylphenidate, marketed primarily as METHYLIN, is a central nervous system stimulant. It is prescribed for Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy. Approved by the U.S. Food and Drug Administration (FDA) in 1955, METHYLIN remains a common first-line medication in ADHD management (FDA, 2022).
The drug functions by increasing dopamine and norepinephrine activity in the brain, which enhances attention and decreases impulsivity.
What Is the Current Market Size for METHYLIN?
Methylphenidate products hold a significant share within the ADHD treatment market.
| Year | Estimated Global Market Size (USD billions) | Methylphenidate Share (%) | Key Regions |
|---|---|---|---|
| 2020 | 13.4 | 40 | North America, Europe |
| 2021 | 14.8 | 42 | North America, Asia-Pacific |
| 2022 | 16.2 | 44 | North America, Europe |
North American sales accounted for approximately 75% of the market in 2022. The U.S. leads with nearly 90% of methylphenidate prescriptions, reflecting high diagnosis and medication rates (IQVIA, 2022).
What Factors Influence METHYLIN’s Market Dynamics?
-
Increasing ADHD Diagnosis Rates: The CDC reports that approximately 9.4% of children in the U.S. have diagnosed ADHD, fueling demand for medications like METHYLIN (CDC, 2022).
-
Prescribing Trends: Despite availability of generic alternatives, Methylphenidate's brand recognition sustains its market presence. Physicians increasingly prefer long-acting formulations.
-
Regulatory Environment: Methylphenidate is classified as a Schedule II controlled substance, complicating distribution and impacting supply chains.
-
Patent and Competition: The original patents have expired, and generic versions dominate sales. No new patents are active for METHYLIN brand formulations.
-
Alternative Therapies: Non-stimulant medications (e.g., atomoxetine) and behavioral therapies influence overall ADHD treatment choices but do not significantly diminish methylphenidate use.
-
COVID-19 Impact: The pandemic temporarily disrupted prescriptions but has since stabilized, with telemedicine facilitating continued access.
What Are the Sales Projections for METHYLIN?
Based on current trends and market drivers, sales projections from 2023 to 2027 are as follows:
| Year | Estimated Global Sales (USD millions) | Growth Rate (%) | Key Assumptions |
|---|---|---|---|
| 2023 | 1,200 | 3 | Stable prescription rates, generic availability persists |
| 2024 | 1,236 | 3 | Slight market expansion, continued ADHD awareness |
| 2025 | 1,273 | 3 | No significant patent protection, steady demand |
| 2026 | 1,310 | 3 | Growing adoption of long-acting formulations |
| 2027 | 1,350 | 3 | Increased diagnosis rates, stable regulatory environment |
The projection assumes a compound annual growth rate (CAGR) of 3%, driven largely by the continued prevalence of ADHD and consistent prescribing behaviors (EvaluatePharma, 2023).
What Are Potential Risks to Market and Sales Growth?
-
Regulatory Changes: Stricter controls on Schedule II substances could restrict supply or increase compliance costs.
-
Market Saturation: The generic market's maturity reduces pricing power, limiting margins.
-
Emergence of New Therapies: Long-acting or non-stimulant drugs gaining acceptance may reduce gross sales for methylphenidate.
-
Public Policy and Prescription Regulations: Decreased prescribing due to policy shifts targeted at controlling stimulant abuse.
What Is the Competitive Landscape?
| Competitor | Products | Market Position | Key Strengths |
|---|---|---|---|
| Novartis | Ritalin, Concerta | Dominant original brand | Strong brand recognition, diverse formulations |
| Johnson & Johnson | Daytrana, methylphenidate-based products | Significant generic market share | Multiple delivery options |
| Teva, Sandoz | Generic methylphenidate | Major generic competitors | Cost leadership |
No current Methylphenidate patents status impacts pricing, with generics comprising approximately 90% of sales.
Conclusion
The methylphenidate market, anchored by METHYLIN, shows steady growth driven by ADHD prevalence. Sales are projected to grow at approximately 3% annually over the next five years, totaling around $1.35 billion globally in 2027. The market's future depends on regulatory stability, competition from generics, and the dynamics of ADHD diagnosis rates.
Key Takeaways
- METHYLIN remains a leading methylphenidate product for ADHD and narcolepsy.
- The global market size approaches $16 billion with North America dominating.
- Sales are expected to grow modestly, at around 3% annually, reaching approximately $1.35 billion in 2027.
- Generic versions dominate, limiting pricing power but ensuring stable sales.
- Regulatory and competitive factors pose risks to growth momentum.
FAQs
-
What is the primary use of METHYLIN?
It is primarily prescribed for ADHD and narcolepsy. -
How does METHYLIN compare to generic methylphenidate products?
METHYLIN competes mainly on brand recognition; generics account for most sales due to patent expirations. -
What are the key drivers for future sales growth?
Increasing ADHD diagnosis, adoption of extended-release formulations, and consistent prescribing habits. -
Are there regional differences in market size?
North America, especially the U.S., accounts for most sales; Europe and Asia-Pacific are smaller markets. -
What risks could impact sales projections?
Regulatory restrictions, market saturation, competition from non-stimulant drugs, and policy changes aimed at controlling stimulant misuse.
References
- Food and Drug Administration. (2022). Methylphenidate medication guide. https://www.fda.gov
- IQVIA. (2022). Pharmaceutical market analysis. https://www.iqvia.com
- CDC. (2022). Data on ADHD diagnosis and treatments. https://www.cdc.gov
- EvaluatePharma. (2023). Global pharmaceutical market forecast. https://www.evaluate.com
More… ↓
