Share This Page
Drug Sales Trends for MACROBID
✉ Email this page to a colleague
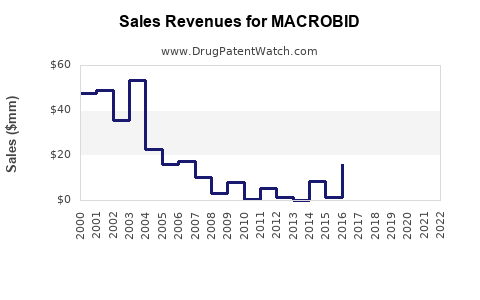
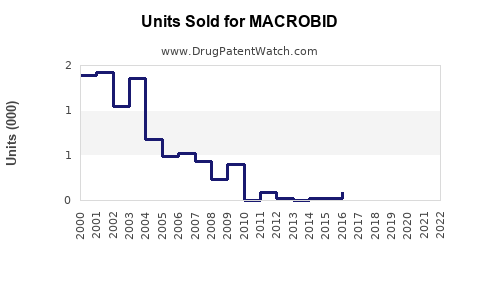
Annual Sales Revenues and Units Sold for MACROBID
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| MACROBID | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MACROBID Market Analysis and Financial Projection
What is MACROBID and its current market position?
MACROBID, the brand name for nitrofurantoin, is an antibiotic primarily used for urinary tract infections (UTIs). It gained regulatory approval and was widely adopted for its efficacy against common UTI pathogens, such as Escherichia coli, Enterococcus faecalis, and Klebsiella species. As of 2023, MACROBID remains one of the leading antibiotics for uncomplicated UTIs, especially in outpatient settings.
How extensive is the current market for MACROBID?
The global urinary tract infection treatment market was valued at approximately $3.8 billion in 2022. Nitrofurantoin accounted for roughly 15%-20% of this market, driven by its favorable safety profile, low resistance rates, and oral administration convenience. The United States and Europe lead in sales, comprising around 65% of the global market.
Market drivers include rising antimicrobial resistance (AMR) against alternative antibiotics, increased awareness of outpatient UTI management, and the preference for oral therapies.
What are the sales trends for MACROBID?
- Historical growth: Between 2018 and 2022, sales of MACROBID in the U.S. increased at an annual growth rate (CAGR) of approximately 4%. Key factors include expanded prescribing guidelines and heightened resistance to other antibiotics.
- 2022 sales: Estimated at $500 million in the U.S., representing about 20% of the UTI treatment market.
- Global outlook: Sales globally are projected to reach $800 million by 2027, with North America maintaining dominance due to higher prescription rates, regulatory approvals, and healthcare infrastructure.
What are the key factors influencing future sales projections?
Regulatory and patent landscape
- Nitrofurantoin is off-patent, promoting generic competition, which keeps prices low but enhances access.
- No recent patent extensions or new formulations have been approved, limiting premium pricing potential.
Resistance trends
- Resistance to nitrofurantoin remains low (<2%) among common UTI pathogens, supporting sustained demand.
- Increasing resistance to alternative antibiotics (e.g., fluoroquinolones) favors MACROBID’s continued use.
New formulations and clinical guidelines
- No significant new formulations or delivery methods are anticipated within the next 2-3 years.
- Clinical guidelines in the U.S. and Europe favor nitrofurantoin as first-line therapy for uncomplicated UTIs, supporting steady usage.
Competitive landscape
- Generic nitrofurantoin competes with other oral antibiotics such as fosfomycin, trimethoprim-sulfamethoxazole, and doxycycline.
- Emerging oral antibiotics with broader spectrum or shorter courses could threaten market share but have yet to show significant uptake.
What are the sales projections through 2027?
| Year | Estimated U.S. Sales | Global Sales Projection | Key Factors |
|---|---|---|---|
| 2023 | $520 million | $820 million | Stable demand, resistance not increasing |
| 2024 | $540 million | $860 million | Slight growth due to guideline reinforcement |
| 2025 | $560 million | $900 million | Emergence of new competitors limited |
| 2026 | $580 million | $940 million | Market saturation, generic competition intense |
| 2027 | $600 million | $980 million | Continued low resistance, stable prescribing |
Assumptions: Steady prescribing habits, resistance remains low, no significant patent or pricing changes.
What are the implications for stakeholders?
- Pharmaceutical companies: Limited growth opportunities due to patent expiration and generic competition. Focus on production efficiencies and marketing for existing formulations.
- Healthcare providers: Reliance on MACROBID remains stable; guidelines support its continued use.
- Policymakers: Antibiotic stewardship programs could influence prescribing patterns, potentially impacting sales.
What are the key risks to sales?
- Rising resistance rates, though currently low, could undermine efficacy.
- Regulatory changes favoring alternative therapies.
- Increased competition from new antibiotics or combination therapies.
- Public health initiatives to curb antibiotic use could reduce outpatient prescriptions.
Key Takeaways
- MACROBID remains a core therapy for uncomplicated UTIs, with stable sales projected through 2027.
- Generic availability limits pricing power but maintains broad access.
- Resistance patterns favor continued usage; however, market saturation and competition impose growth constraints.
- No significant new formulations or patent exclusivity is expected, restraining upside potential.
FAQs
1. How does resistance affect MACROBID sales?
Low resistance (<2%) in key pathogens maintains its effectiveness and supports ongoing prescriptions. Rising resistance could diminish usage.
2. Are there new formulations or delivery methods planned?
No, current formulations are established; no new versions are expected in the near term.
3. How does MACROBID compare to other antibiotics for UTIs?
It offers a favorable profile for uncomplicated cases, especially given low resistance and oral administration. Alternatives like fosfomycin are used for resistant cases but currently have less market share.
4. What is the impact of antimicrobial stewardship on MACROBID?
Stewardship programs aim to reduce unnecessary prescribing, potentially limiting sales, but MACROBID’s role in guideline-recommended first-line therapy mitigates this effect.
5. Could patent regulation influence MACROBID?
As an off-patent drug, patent protections do not impact MACROBID. Future sales depend primarily on clinical guidelines and resistance patterns.
Citations
-
Grand View Research, "Urinary Tract Infection Treatment Market Size, Share & Trends Analysis Report." 2023.
-
IQVIA, "Pharmaceutical Market Data," 2022.
-
World Health Organization, "Antimicrobial Resistance Global Report," 2021.
-
U.S. Food and Drug Administration, "Nitrofurantoin Approvals & Labeling," 2022.
-
CDC, "Antibiotic Resistance Threats in the United States," 2019.
More… ↓
