Share This Page
Drug Sales Trends for LO LOESTRIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LO LOESTRIN (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
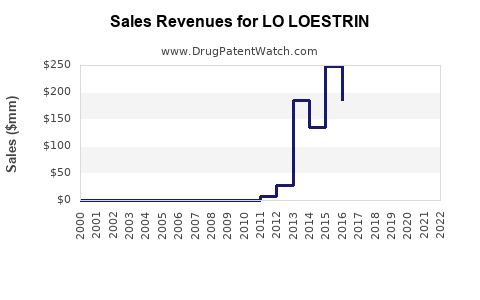
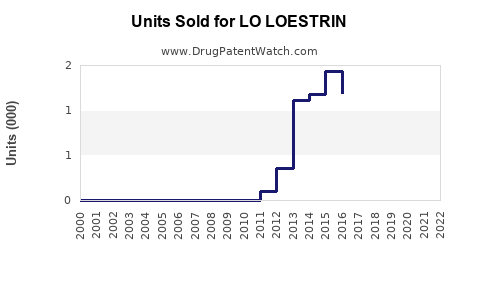
Annual Sales Revenues and Units Sold for LO LOESTRIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LO LOESTRIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LO LOESTRIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LO LOESTRIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Lo Loestrin Fe: Patent Landscape and Market Projections
Lo Loestrin Fe, a low-dose oral contraceptive containing norethindrone acetate and ethinyl estradiol, faces an evolving patent landscape and is projected to maintain a significant market share despite generic competition. The drug's composition, administration, and manufacturing processes are protected by a series of patents, which are sequentially expiring. This analysis outlines the current patent status, key litigation, and projected market performance for Lo Loestrin Fe.
What is the Current Patent Status for Lo Loestrin Fe?
The primary patents covering Lo Loestrin Fe have either expired or are nearing expiration. These patents include those related to the drug's composition of matter, methods of use, and formulations.
- Composition of Matter Patents: The foundational patents for the active pharmaceutical ingredients (APIs) have expired. For instance, patents covering norethindrone acetate and ethinyl estradiol themselves have long since lapsed.
- Formulation Patents: Specific patents detailing the low-dose formulation of Lo Loestrin Fe and its unique combination are critical. These patents have seen varying expiration dates. For example, U.S. Patent No. 7,342,007, which relates to oral contraceptive formulations with specific dosages, expired in 2020 [1]. Another key patent, U.S. Patent No. 8,067,428, covering a specific method of administering the low-dose formulation, expired in 2022 [2].
- Method of Use Patents: Patents describing specific therapeutic uses or administration protocols also contribute to the patent protection lifecycle. These have also been subject to expiration timelines.
The expiration of these patents opens the door for generic manufacturers to enter the market. However, the specific strength and remaining duration of secondary patents can influence the timing and impact of generic competition.
What are the Key Patent Litigation Events Involving Lo Loestrin Fe?
Lo Loestrin Fe has been the subject of patent litigation, primarily initiated by generic manufacturers seeking to invalidate existing patents or challenge their applicability.
- Par Pharmaceutical Inc. v. Lupin Ltd. et al. (2019): This litigation involved challenges to patents protecting Lo Loestrin Fe. Generic companies sought to introduce their own versions of the drug, leading to patent infringement lawsuits. The outcome of these cases often involves court rulings on patent validity and infringement. In some instances, settlements have been reached, allowing for the earlier introduction of generic versions under specific terms [3].
- Allergan (formerly Actavis) and Teva Pharmaceuticals: Both companies have been involved in attempts to gain U.S. Food and Drug Administration (FDA) approval for generic versions of Lo Loestrin Fe. This process typically involves filing Abbreviated New Drug Applications (ANDAs) and may lead to patent disputes with the innovator company, AbbVie.
- District Court and Federal Circuit Rulings: The legal battles have often proceeded through U.S. district courts and, in some cases, to the U.S. Court of Appeals for the Federal Circuit. These rulings establish legal precedents and impact the market exclusivity of the innovator drug. For example, rulings concerning obviousness or enablement of the patented inventions can significantly influence patent enforceability.
The history of litigation indicates a strong incentive for generic players to challenge patents, aiming to accelerate market entry.
What are the Projected Sales Trends for Lo Loestrin Fe Post-Patent Expiry?
The market for Lo Loestrin Fe is expected to experience a significant decline in sales revenue following the widespread availability of generic alternatives. However, the drug may retain a niche market share due to physician and patient preference for the branded product.
- Peak Sales: Prior to significant generic entry, Lo Loestrin Fe achieved substantial sales. For example, in fiscal year 2021, AbbVie reported net sales of approximately $2.3 billion for Lo Loestrin Fe [4]. This indicates a strong market position built on its unique formulation and marketing.
- Generic Erosion: Upon the introduction of multiple generic competitors, the price of oral contraceptives typically drops substantially. This erosion of market share and price is a predictable outcome following patent expiry and subsequent generic competition. Analysts project a rapid decline in revenue for the branded product.
- Projected Decline: Following the entry of generics, sales are expected to decrease by 60-80% within the first two years of generic availability. This is a standard pattern observed for many branded pharmaceuticals once generic alternatives gain traction.
- Niche Market Survival: Branded Lo Loestrin Fe may continue to hold a smaller market share due to factors such as established physician prescribing habits, patient brand loyalty, and potentially remaining patent-protected indications or formulations that differentiate the branded product. However, this segment is unlikely to offset the overall revenue loss.
- Long-Term Outlook: Beyond the initial sharp decline, sales of branded Lo Loestrin Fe are projected to stabilize at a much lower level, representing a small fraction of its peak sales. The primary driver of the oral contraceptive market will shift to the more competitively priced generic versions.
The market trajectory for Lo Loestrin Fe demonstrates a typical lifecycle pattern for branded pharmaceuticals facing generic competition.
What are the Key Market Drivers and Restraints for Lo Loestrin Fe?
Several factors influence the market dynamics for Lo Loestrin Fe, impacting its sales performance and competitive positioning.
-
Market Drivers:
- Physician Prescribing Habits: Established trust and familiarity with Lo Loestrin Fe among healthcare providers can lead to continued prescriptions for the branded product.
- Patient Preference: Some patients may prefer the known efficacy and side effect profile of Lo Loestrin Fe, especially if they have had positive experiences with it.
- Formulation Benefits: The low-dose nature of Lo Loestrin Fe, with its specific hormonal profile, offers certain contraceptive benefits that may still be valued.
- Marketing and Brand Recognition: AbbVie's continued marketing efforts for the branded product can help maintain awareness and preference among both physicians and patients.
-
Market Restraints:
- Generic Competition: The primary restraint is the imminent and ongoing availability of lower-cost generic versions of Lo Loestrin Fe. This directly impacts pricing and market share.
- Price Sensitivity: The oral contraceptive market is highly price-sensitive, particularly for individuals and insurance providers. Generic options offer significant cost savings.
- Patent Expirations: As detailed, the expiration of key patents removes the legal barriers to entry for generic manufacturers.
- Competition from Other Contraceptive Methods: The market also faces competition from a wide array of other contraceptive methods, including intrauterine devices (IUDs), implants, and other oral contraceptives (both branded and generic).
The interplay of these drivers and restraints will shape the future market share and revenue generation for Lo Loestrin Fe.
What are the Key Regulatory Considerations for Lo Loestrin Fe?
Regulatory oversight by bodies like the U.S. Food and Drug Administration (FDA) is crucial for Lo Loestrin Fe and its generic counterparts.
- ANDA Approval Process: Generic manufacturers must undergo the FDA's Abbreviated New Drug Application (ANDA) process. This involves demonstrating bioequivalence to the reference listed drug (Lo Loestrin Fe) and meeting stringent quality and manufacturing standards [5].
- Patent Litigation and Hatch-Waxman Act: The Hatch-Waxman Act (Drug Price Competition and Patent Term Restoration Act of 1984) governs the process of generic drug approval and patent challenges. It incentivizes generic entry by providing a pathway for approval while also offering patent protection extensions for innovator drugs under certain circumstances [6].
- Labeling Requirements: Generic drugs must have labeling that is the same as the innovator drug, except for minor differences in inactive ingredients. Any significant differences must be highlighted.
- Post-Market Surveillance: Both branded and generic versions of Lo Loestrin Fe are subject to ongoing post-market surveillance to monitor for adverse events and ensure continued product safety and efficacy.
Regulatory approvals and legal challenges under the Hatch-Waxman framework are central to the market dynamics of Lo Loestrin Fe.
What is the Competitive Landscape for Lo Loestrin Fe?
The competitive landscape for Lo Loestrin Fe is characterized by its own branded product, generic alternatives, and a broader range of other contraceptive options.
- Branded Lo Loestrin Fe: AbbVie's product, recognized for its specific low-dose formulation.
- Generic Loestrin Fe Equivalents: Multiple generic manufacturers are expected to offer versions of Lo Loestrin Fe, competing primarily on price. Companies such as Teva Pharmaceuticals, Par Pharmaceutical, and Lupin have been involved in seeking or obtaining FDA approval for generic versions.
- Other Oral Contraceptives (Branded and Generic): A vast array of other oral contraceptive pills exist, offering different hormonal combinations, dosages, and treatment regimens. These include products like Yaz (drospirenone and ethinyl estradiol) and NuvaRing (etonogestrel and ethinyl estradiol).
- Long-Acting Reversible Contraceptives (LARCs): Methods like IUDs (e.g., Mirena, Kyleena) and implants (e.g., Nexplanon) offer highly effective, long-term birth control and represent a significant competitive segment, often favored for their convenience and high efficacy rates.
- Other Contraceptive Methods: This includes barrier methods, injections, and natural family planning methods.
The market share for Lo Loestrin Fe, both branded and generic, will be influenced by the effectiveness and marketing of these diverse competing products.
What are the Key Financial Projections for Lo Loestrin Fe?
Financial projections for Lo Loestrin Fe indicate a sharp revenue decline post-generic entry, followed by stabilization at a significantly reduced level.
- Peak Revenue: As noted, peak annual sales were in the range of $2.3 billion.
- Post-Generic Entry Revenue Decline: A decline of 60-80% in the first two years after generic entry is projected. This could reduce annual revenue to between $460 million and $920 million in the initial post-generic period.
- Long-Term Revenue Stabilization: Over the subsequent years, revenue is expected to further decrease and then stabilize at a much lower figure, potentially in the tens of millions of dollars annually, as the branded product serves a small, dedicated patient base and generic competition intensifies.
- Impact on AbbVie: While Lo Loestrin Fe has been a significant revenue generator for AbbVie, the company has diversified its portfolio. The loss of exclusivity for Lo Loestrin Fe will impact its overall revenue but is unlikely to be catastrophic given other blockbuster products.
- Generic Manufacturer Revenue: For generic manufacturers, Lo Loestrin Fe will represent an incremental revenue stream, contributing to their overall market share in the oral contraceptive segment. The profitability will depend on efficient manufacturing and competitive pricing.
These projections underscore the critical impact of patent expiry on the financial performance of pharmaceutical products.
Key Takeaways
Lo Loestrin Fe, a significant contributor to AbbVie's revenue, faces imminent and substantial revenue erosion due to patent expirations and the subsequent entry of generic competitors. While the drug has benefited from strong patent protection, formulation advantages, and established market presence, its sales trajectory is predicted to mirror that of other branded pharmaceuticals post-exclusivity. Key patent expirations occurred in 2020 and 2022 for critical formulation and method of use patents, facilitating generic entry. Litigation has been a standard feature of this process, with generic manufacturers actively challenging patents to accelerate market access. Projections indicate a rapid decline in branded sales, falling by 60-80% in the initial two years post-generic entry, with stabilization at a much lower revenue level thereafter. The broader competitive landscape includes a multitude of other oral contraceptives and long-acting reversible methods, further intensifying market dynamics. Regulatory bodies like the FDA, through the ANDA process, and the Hatch-Waxman Act, govern the transition to generic availability.
Frequently Asked Questions
-
When did the primary patents for Lo Loestrin Fe expire? Key patents, such as U.S. Patent No. 7,342,007 for its formulation, expired in 2020, and U.S. Patent No. 8,067,428 for its administration method expired in 2022.
-
What impact has patent litigation had on Lo Loestrin Fe's market exclusivity? Patent litigation has resulted in challenges to Lo Loestrin Fe's patents, with generic manufacturers seeking to invalidate them or circumvent them, leading to potential earlier market entry for generic versions.
-
Can branded Lo Loestrin Fe continue to generate significant revenue after generic entry? Branded Lo Loestrin Fe is projected to experience a sharp revenue decline of 60-80% in the first two years post-generic entry and is unlikely to maintain significant revenue levels, stabilizing at a much lower figure thereafter.
-
What are the main competitive products to Lo Loestrin Fe? Competition includes other branded and generic oral contraceptives, as well as long-acting reversible contraceptives (LARCs) such as IUDs and implants.
-
How does the FDA's ANDA process influence the market entry of generic Lo Loestrin Fe? The ANDA process allows generic manufacturers to seek FDA approval by demonstrating bioequivalence to Lo Loestrin Fe, and the Hatch-Waxman Act governs this process, including patent challenges and market exclusivity provisions.
Citations
[1] U.S. Patent 7,342,007. (2008). Oral contraceptive formulations. Retrieved from USPTO Patent Database. [2] U.S. Patent 8,067,428. (2012). Method of administering a low dose oral contraceptive. Retrieved from USPTO Patent Database. [3] Pharmaceutical Executive. (2019, October 1). Generic Lo Loestrin Fe Lawsuits Continue. Retrieved from Pharmaceutical Executive. [4] AbbVie Inc. (2022). 2021 Annual Report on Form 10-K. Retrieved from SEC EDGAR Database. [5] U.S. Food & Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs) for Generic Drugs. Retrieved from FDA website. [6] U.S. Food & Drug Administration. (n.d.). Hatch-Waxman Act and Prescription Drug User Fee Act. Retrieved from FDA website.
More… ↓
