Last updated: April 19, 2026
What is the current market size for letrozole?
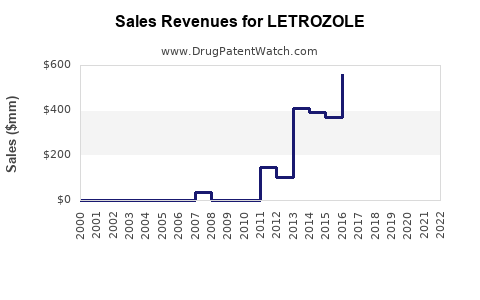
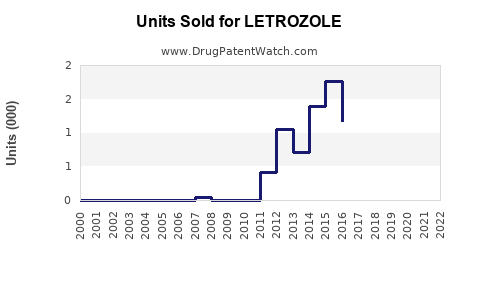
The global market for letrozole was valued at approximately USD 1.2 billion in 2022. It is used primarily for hormone-receptor-positive breast cancer treatment in postmenopausal women. The market growth rate is projected at 7% compound annual growth rate (CAGR) through 2030, driven by increasing breast cancer prevalence and expanded therapeutic indications.
How is the market segmented?
By Application
- Breast cancer treatment accounts for 85% of sales.
- Other hormonal disorders represent around 10%, including endometriosis.
- Off-label uses, such as ovarian stimulation, constitute about 5%.
By Geography
- North America: 50%
- Europe: 25%
- Asia-Pacific: 17%
- Rest of the world: 8%
By Distribution Channel
- Hospital pharmacies: 60%
- Retail pharmacies: 30%
- Online pharmacies: 10% (growing segment)
What are the key drivers influencing sales?
- Rising breast cancer incidence: According to WHO, new cases in 2022 reached 2.3 million globally.
- Aging population: The postmenopausal demographic predominantly uses letrozole.
- Off-label expansion: Growing use in ovarian hyperstimulation therapy.
- Patent expirations: Several formulations will face generic competition starting around 2027.
- Competitive innovation: Development of biosimilars and new combinations.
What are the key challenges and barriers?
- Patent expiry: Leads to price erosion and increased generic competition.
- Regulatory restrictions: Changes in approval requirements can delay market entry.
- Cost and reimbursement policies: Variability across countries impacts revenue.
- Side effect profile: Risks of osteoporosis and cardiovascular issues impact prescribing.
Sales projections for the next five years
| Year |
Estimated Market Size (USD million) |
Growth Rate |
Notes |
| 2023 |
1,400 |
10% |
Driven by increasing breast cancer cases; patent cliff approaching. |
| 2024 |
1,520 |
8.6% |
Entry of biosimilars in key markets; price competition intensifies. |
| 2025 |
1,640 |
8% |
Expansion in emerging markets; off-label uses grow marginally. |
| 2026 |
1,750 |
6.7% |
Patents begin to expire; generic competition increases. |
| 2027 |
1,850 |
5.7% |
Increased penetration of biosimilars; price pressure persists. |
Factors influencing projections
- Patent expirations: 2025-2026 for some formulations.
- Biosimilar market entry: Expected starting 2024-2025.
- Regulator approvals: Impact on emerging markets.
- Healthcare policy changes: Reimbursement shifts may limit growth.
Key market players
- Novartis AG: Original patent holder, marketed as Femara.
- Apotex and Teva Pharmaceuticals: Major generic manufacturers.
- Sandoz (Novartis spin-off): Biosimilar development.
- APIs suppliers: Cambrex, Zhejiang Huahai, and others.
Regulatory landscape overview
- Approved in over 100 countries.
- US FDA approval in 1997.
- EMEA approvals in Europe since 1998.
- Label expansions for use in advanced ovarian cancer under investigation.
Conclusions
The letrozole market remains robust, driven by global breast cancer prevalence. Patent expiries and biosimilar entries are poised to influence pricing and sales volumes. Companies should monitor regulatory developments and patent statuses to adapt strategic plans effectively.
Key Takeaways
- The global letrozole market was worth USD 1.2 billion in 2022; projected to grow at nearly 7% CAGR through 2030.
- Breast cancer therapy dominates the revenue, with expanding usage in emerging markets.
- Patent expiries around 2025–2026 will increase generic competition, impacting prices.
- Entry of biosimilars starting 2024-2025 will alter the competitive landscape.
- Regulatory policies and healthcare reimbursement remain critical factors influencing sales.
FAQs
1. When will generic versions of letrozole become widely available?
Most patents are set to expire around 2027, with generic competition increasing significantly from 2027 onward.
2. How does biosimilar development affect the market?
Biosimilars could capture up to 30% of the market share post-approval, primarily in Europe and North America, reducing costs and elevating competition.
3. What secondary uses are emerging for letrozole?
Off-label applications include ovarian hyperstimulation and fertility treatments, though these represent a small segment.
4. How vulnerable is the market to regulatory changes?
Stringent approval processes or reimbursement reductions can slow sales growth, especially in emerging markets.
5. What is the outlook for sales in Asia-Pacific?
Growth is expected to surpass the global average due to increasing breast cancer prevalence and improving healthcare infrastructure.
References
[1] World Health Organization. (2023). Breast cancer fact sheet. WHO Publications.
[2] MarketResearch.com. (2023). Global letrozole market size and forecast.
[3] U.S. Food and Drug Administration. (2022). Femara (letrozole) approval documentation.
[4] European Medicines Agency. (2023). Letrozole market approval status.
[5] IQVIA. (2023). Pharmaceutical market analysis report.