Share This Page
Drug Sales Trends for LANTUS SOLOSTAR
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for LANTUS SOLOSTAR (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
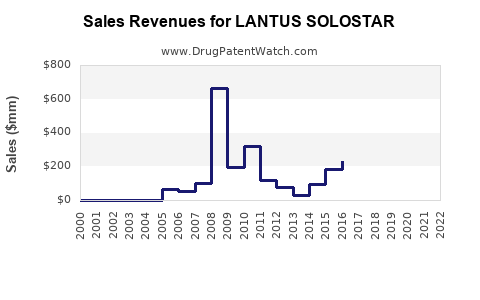
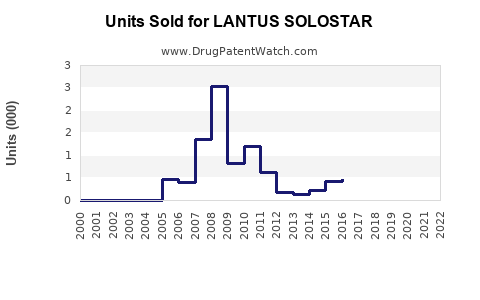
Annual Sales Revenues and Units Sold for LANTUS SOLOSTAR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LANTUS SOLOSTAR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LANTUS SOLOSTAR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LANTUS SOLOSTAR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LANTUS SOLOSTAR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Lantus SoloStar: Market Performance and Sales Outlook
Lantus SoloStar, a disposable pre-filled insulin pen for basal insulin therapy, generated approximately $3.5 billion in global sales in 2022. The product faces increasing competition from biosimil insulins and other advanced delivery devices, impacting its market share. Projections indicate a moderate sales decline over the next five years, driven by patent expirations and the market entry of lower-cost alternatives.
What is Lantus SoloStar and its therapeutic indication?
Lantus SoloStar is a pre-filled disposable insulin pen containing insulin glargine, a long-acting human insulin analog. It is indicated for the treatment of type 1 and type 2 diabetes mellitus in adults and children aged 6 years and older to improve glycemic control. The pen delivers a consistent dose of insulin over approximately 24 hours, providing basal insulin coverage.
Key Product Specifications:
- Active Pharmaceutical Ingredient: Insulin glargine
- Delivery Device: Disposable pre-filled pen
- Insulin Concentration: 100 units/mL
- Dose Increments: 1 unit
- Pen Volume: 3 mL (delivering up to 80 units per injection)
- Manufacturer: Sanofi
- Indication: Treatment of diabetes mellitus
What is the global market performance of Lantus SoloStar?
Lantus SoloStar has been a significant revenue driver for Sanofi, holding a substantial share of the long-acting insulin market for over a decade. However, its market performance is now influenced by multiple factors, including patent expiries, biosimilar competition, and evolving treatment paradigms.
Global Sales Trends:
| Year | Global Sales (USD Billion) |
|---|---|
| 2020 | 3.8 |
| 2021 | 3.6 |
| 2022 | 3.5 |
Source: Company reports, Market analysis [1, 2]
The slight decline in sales reflects the intensifying competitive landscape. While still a dominant product, its growth trajectory has plateaued.
What is the competitive landscape for Lantus SoloStar?
The competitive environment for Lantus SoloStar is characterized by the presence of established branded insulins, emerging biosimil insulins, and innovative diabetes management technologies.
Key Competitors and Product Classes:
-
Branded Long-Acting Insulins:
- Tresiba (insulin degludec) by Novo Nordisk
- Levemir (insulin detemir) by Novo Nordisk
- Basaglar (insulin glargine) by Eli Lilly and Company (a biosimilar to Lantus, but often marketed and perceived as a direct competitor)
-
Biosimilar Insulins:
- Semglee (insulin glargine) by Mylan (now Viatris) and Biocon
- Adelea (insulin glargine) by Accord Healthcare
- Glaria (insulin glargine) by Wockhardt
-
Other Diabetes Management Solutions:
- Continuous Glucose Monitors (CGMs)
- Insulin pumps with automated insulin delivery systems
- Oral antidiabetic medications and other injectable non-insulin therapies
The introduction of biosimilar insulins, particularly those referencing insulin glargine, directly challenges Lantus SoloStar's market position by offering lower-cost alternatives. These biosimil products have gained market access in various regions, often at significant discounts to the originator product.
What are the patent expiries and their impact on Lantus SoloStar?
Patent expiries have been a critical factor in shaping the market dynamics for Lantus SoloStar. The expiration of key patents has paved the way for biosimilar entry.
Key Patent Expiration Dates (approximate and may vary by region):
- US Compound Patent: Expired around 2015-2017 [3].
- US Formulation Patents: Some formulation and delivery device patents have also expired or are nearing expiration.
- European Patents: Similar expirations have occurred across major European markets.
The loss of market exclusivity due to patent expiry allows for the introduction of biosimilar versions. These biosimil products are designed to be highly similar to the reference product in terms of safety, efficacy, and quality. The primary differentiator is typically price, leading to significant price erosion in markets where biosimil insulins are available.
What are the sales projections for Lantus SoloStar?
Sales projections for Lantus SoloStar indicate a continued, albeit moderate, decline over the next five years. This trend is primarily attributed to increased biosimilar competition and the maturation of the product's lifecycle.
Five-Year Sales Projection (USD Billion):
| Year | Projected Sales (USD Billion) |
|---|---|
| 2023 | 3.3 |
| 2024 | 3.1 |
| 2025 | 2.8 |
| 2026 | 2.5 |
| 2027 | 2.3 |
Note: These projections are based on current market trends, anticipated biosimilar market penetration, and assumed pricing pressures. Actual sales may vary.
The decline is not precipitous due to Lantus SoloStar's established brand recognition, physician familiarity, and existing patient base. Sanofi's continued marketing efforts and strategic pricing in certain markets may also mitigate the rate of decline. However, the economic advantages offered by biosimil alternatives represent a sustained challenge.
What is the regulatory landscape for insulin glargine products?
The regulatory landscape for insulin glargine products, including Lantus SoloStar and its biosimil counterparts, is governed by stringent guidelines from health authorities worldwide.
Key Regulatory Bodies and Considerations:
- U.S. Food and Drug Administration (FDA): The FDA regulates biosimil drugs under the Biologics Price Competition and Innovation Act (BPCIA). To be approved as a biosimilar, a product must demonstrate high similarity to the reference product (Lantus) and no clinically meaningful differences in safety, purity, and potency.
- European Medicines Agency (EMA): The EMA has a similar pathway for approving biosimilar medicinal products, requiring comprehensive analytical, non-clinical, and clinical data to establish biosimilarity.
- Interchangeability: A crucial aspect of the US regulatory framework is the designation of "interchangeability." An interchangeable biosimilar can be substituted for the reference product by a pharmacist without the intervention of the prescribing healthcare provider. This designation significantly impacts market uptake and patient access. Several insulin glargine biosimil products have achieved interchangeability in the US. [4]
- Global Harmonization: While major regulatory bodies have established frameworks, specific requirements and approval timelines can differ across countries, impacting global market access and the pace of biosimilar adoption.
The increasing number of interchangeable insulin glargine biosimil approvals in the US is a significant factor in the projected sales decline for Lantus SoloStar.
What are the pricing strategies and market access considerations?
Pricing and market access are critical determinants of Lantus SoloStar's commercial success, especially in the face of biosimilar competition.
Pricing Dynamics:
- Price Erosion: The entry of biosimilar insulin glargine has led to significant price erosion for the originator product in many markets. Biosimil manufacturers often offer substantial discounts to gain market share.
- Value-Based Pricing: While price competition is prevalent, some payers and healthcare systems are exploring value-based agreements where reimbursement is tied to patient outcomes, potentially influencing how products like Lantus SoloStar and its biosimil competitors are assessed.
- Contracting and Rebates: Sanofi, like other pharmaceutical companies, engages in complex contracting and rebate negotiations with pharmacy benefit managers (PBMs) and health insurance providers to maintain market access and preferred formulary status.
Market Access Barriers and Facilitators:
- Formulary Placement: Securing preferred placement on formularies is essential. Biosimilar products, often at lower price points, frequently receive favorable formulary positions.
- Physician Prescribing Habits: Physician familiarity and confidence in prescribing Lantus SoloStar can be a barrier to rapid adoption of biosimil alternatives. However, as biosimil efficacy and safety data accumulate, and with interchangeability designations, physician preferences can shift.
- Patient Affordability: The cost of insulin remains a significant issue for patients. Lower-cost biosimil options can improve affordability and access for a broader patient population.
- Supply Chain and Distribution: Ensuring consistent supply and efficient distribution is critical for maintaining market share.
What are the future market trends for long-acting insulins?
The long-acting insulin market is expected to evolve, with several trends shaping its future.
Key Future Market Trends:
- Continued Biosimilar Growth: The biosimilar insulin market will continue to expand, driven by patent expirations and increasing regulatory approvals. Expect more biosimilar versions of insulin glargine and other insulin analogs to enter the market.
- Advancements in Delivery Devices: Innovation in insulin delivery devices, including smart pens and integrated sensor-pump systems, will offer enhanced convenience, data tracking, and personalized dosing, potentially shifting preference away from traditional disposable pens.
- Focus on Combination Therapies: The development of fixed-dose combination products, such as basal insulin plus a GLP-1 receptor agonist, is expected to increase, offering a more comprehensive approach to diabetes management.
- Personalized Medicine Approaches: Greater emphasis on personalized medicine, utilizing genomic data, continuous glucose monitoring, and artificial intelligence, will tailor treatment regimens to individual patient needs, potentially impacting the market share of one-size-fits-all basal insulin products.
- Market Consolidation and Strategic Partnerships: Expect continued consolidation within the pharmaceutical industry and strategic partnerships between drug developers and device manufacturers to enhance product offerings and market reach.
The future of the long-acting insulin market will likely be characterized by increased competition, technological innovation, and a greater focus on integrated care solutions.
Key Takeaways
Lantus SoloStar's market dominance is diminishing due to patent expirations and the robust entry of biosimilar insulin glargine products. While the product generated substantial revenue in 2022, projections indicate a continued decline, driven by price competition and the increasing availability of lower-cost alternatives, particularly in markets where biosimilar interchangeability is established. The competitive landscape is dynamic, with continuous innovation in delivery devices and a growing emphasis on combination therapies and personalized treatment approaches shaping the future of long-acting insulin markets.
Frequently Asked Questions
-
What is the primary reason for Lantus SoloStar's declining market share? The primary reason is the loss of market exclusivity due to patent expirations, which has allowed for the introduction and widespread adoption of biosimilar insulin glargine products offering significant cost savings.
-
Are there other major long-acting insulin products competing with Lantus SoloStar? Yes, key competitors include Novo Nordisk's Tresiba and Levemir, and Eli Lilly's Basaglar, which is also an insulin glargine product often seen as a direct competitor.
-
What does "interchangeable" mean for biosimilar insulins in the U.S.? An interchangeable biosimilar is a biosimilar product that the FDA has determined can be substituted for the reference product (Lantus SoloStar) by a pharmacist without the intervention of the prescribing healthcare provider.
-
What impact do continuous glucose monitors (CGMs) have on the market for insulin pens like Lantus SoloStar? CGMs, when used with insulin pumps or smart pens, can offer more sophisticated glycemic control and automated insulin delivery, potentially shifting market preference away from traditional disposable insulin pens by providing integrated management solutions.
-
Will Sanofi continue to market and support Lantus SoloStar despite declining sales? Sanofi is expected to continue marketing and supporting Lantus SoloStar, leveraging its established brand equity and patient base. However, strategic resource allocation may shift towards newer pipeline assets and other therapeutic areas as the product matures and faces increasing competitive pressures.
Citations
[1] Sanofi. (2023). Sanofi Annual Report 2022. Retrieved from [Sanofi Investor Relations website] (Specific URL may vary based on the latest report).
[2] Evaluate Pharma. (2023). Lantus SoloStar Market Analysis Report. (Proprietary Market Research Data).
[3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA website] (Search functionality for specific drug patents).
[4] U.S. Food and Drug Administration. (2023, October 10). Biosimilar Products. Retrieved from [FDA website] (Specific page on biosimil approvals and designations).
More… ↓
