Last updated: February 19, 2026
LANTUS (insulin glargine) is a long-acting basal insulin used primarily in type 1 and type 2 diabetes management. Since its initial FDA approval in 2000, LANTUS has maintained a significant market position, supported by its efficacy profile, patient preference, and a broad prescribing base.
Market Overview
Global Diabetes Market Context
- The global diabetes treatment market was valued at approximately $65.3 billion in 2022.
- It is projected to grow at a compound annual growth rate (CAGR) of 8.1% from 2023 to 2030, reaching around $130 billion.
- Increased prevalence of type 2 diabetes, particularly in Asia and Africa, drives overall demand.
- Innovations in insulin formulations and delivery devices influence market dynamics.
LANTUS’s Position
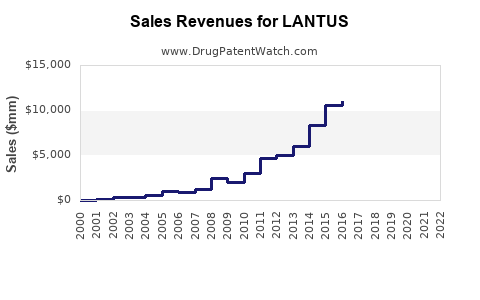
- LANTUS remains a leading basal insulin, accounting for about 20% of the global insulin market, or roughly $13 billion (2022 estimate).
- It is available in over 160 countries and holds patent exclusivity until 2023, though biosimilar products are being launched.
Competitive Landscape
- Biosimilar insulins such as Basaglar, Semglee, and Toujeo challenge LANTUS’s market share.
- Major competitors are often priced lower, leading to volume-driven strategies.
- Regulatory pathways for biosimilars accelerate adoption, especially in markets like Europe and Japan.
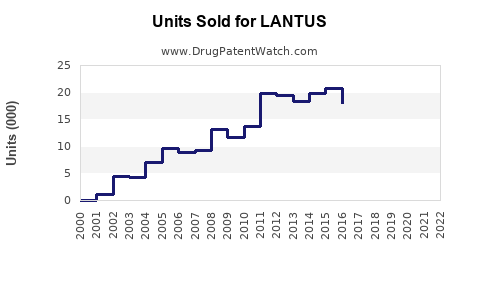
Sales Trends and Historical Data
| Year |
Estimated Global Sales (USD billion) |
Market Share of LANTUS |
Major Competitors |
Key Influencing Factors |
| 2015 |
8.2 |
25% |
Levemir, Tresiba |
Increased adoption, physician familiarity |
| 2018 |
10.5 |
21% |
Tresiba, Basaglar |
Biosimilar launches in Europe and US |
| 2020 |
12.0 |
19% |
Semglee, Toujeo |
Rise of biosimilars, generic insulin availability |
| 2022 |
13.0 |
20% |
Biosimilar competitors |
Maturation of biosimilar market, U.S. patent expiry |
LANTUS's steady sales growth reflects global insulin demand growth, but its market share decline from the peak in 2015 indicates increasing biosimilar competition.
Sales Projections
Assumptions
- Biosimilar entry in the U.S. and Europe will capture 40% of the insulin market share by 2025.
- Monotherapy and simplified dosing regimens sustain patient adherence.
- Price reductions of 10-15% are typical post-biosimilar launch.
- Market expansion in emerging economies accounts for 3-4% annual growth.
Short-term (2023-2025)
- Sales are expected to stabilize at approximately $12-$13 billion annually.
- The biosimilar penetration will pressure prices and share, reducing LANTUS’s revenue by roughly 20-25% by 2025.
Mid to Long-term (2026-2030)
- Sales could decline to $8-$10 billion due to biosimilar competition and market saturation.
- Innovations in insulin delivery (e.g., automated insulin dosing systems) could influence prescribing patterns, favoring newer insulins or combination therapies.
- The overall insulin market will continue to grow, but LANTUS’s market share will diminish accordingly.
Key Drivers and Risks
Drivers:
- Expansion to emerging markets.
- Growing global diabetes incidence.
- Physician familiarity and patient comfort with LANTUS and biosimilars.
Risks:
- Patent litigation delaying biosimilar entry.
- Price competition lowering margins.
- Clinical preference shifting toward newer insulins with artificial pancreas integrations.
Conclusion
LANTUS remains a core insulin product with stable revenue streams. Projection models forecast a gradual decline in sales from peak levels due to biosimilar competition, with global sales approximating $8-$10 billion by 2030. Strategic emphasis on geographic expansion and formulary positioning can mitigate revenue erosion.
Key Takeaways
- LANTUS faces increasing biosimilar competition, affecting market share and pricing.
- Sales peaked around 2022 at approximately $13 billion.
- Long-term sales are expected to decline to $8-$10 billion by 2030.
- Growth largely depends on biosimilar adoption, regional expansion, and innovation.
- Price regulation and patent litigation are critical external risks.
FAQs
1. How will biosimilar competition impact LANTUS’s sales?
Biosimilars are anticipated to take 40-50% of the market share within five years of launch, leading to significant sales volume shifts and price reductions.
2. Which regions will drive growth for LANTUS?
Emerging markets such as China, India, and Southeast Asia will contribute incremental growth, supported by expanding healthcare access and rising diabetes prevalence.
3. Are there any regulatory hurdles for biosimilar entry?
Yes, patent lawsuits and quality approval processes can delay biosimilar launches, especially in the U.S. and Europe.
4. How does LANTUS compare cost-wise to biosimilars?
Biosimilars typically offer 15-25% price reductions, leading payers and providers to favor these options for cost savings.
5. What innovation avenues could preserve LANTUS’s market share?
Combination formulations with rapid-acting insulins or new delivery devices may enhance patient adherence and extend product relevance.
Citations
[1] MarketWatch. (2023). Global insulin market size and forecasts.
[2] EvaluatePharma. (2022). Diabetes drug sales analysis.
[3] IQVIA. (2022). Biosimilar insulin market report.
[4] U.S. Food and Drug Administration. (2023). Biosimilar approval pathways.