Share This Page
Drug Sales Trends for IPRATROPIUM BROMIDE
✉ Email this page to a colleague
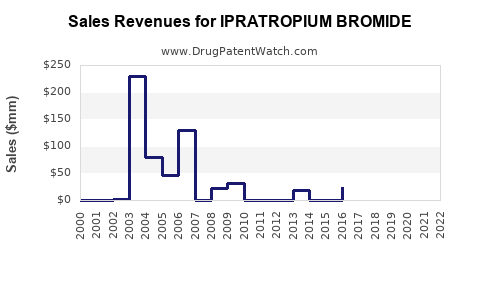
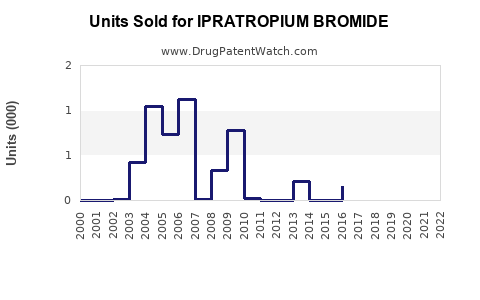
Annual Sales Revenues and Units Sold for IPRATROPIUM BROMIDE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| IPRATROPIUM BROMIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| IPRATROPIUM BROMIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| IPRATROPIUM BROMIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market analysis and sales projections for ipratropium bromide
Ipratropium bromide remains a significant player in respiratory therapeutics, particularly for COPD and asthma management. The global market for inhaled anticholinergic drugs, including ipratropium bromide, demonstrates steady growth driven by high prevalence of respiratory diseases and aging populations.
Market Overview
Current market size (2022):
The global inhalation drugs market was valued at approximately $19 billion in 2022, with anticholinergic agents constituting roughly 30% of this segment, equating to $5.7 billion. Ipratropium bromide specifically accounts for about 70-80% of inhaled anticholinergics used in COPD treatment.
Geographical distribution:
North America leads with 40% market share, followed by Europe with 25%. Asia-Pacific exhibits rapid growth, expected to increase at a CAGR of 7% through 2027. Growth is propelled by rising COPD prevalence, improved healthcare access, and increased awareness.
Key competitors:
- Merck (brand: Atrovent)
- Boehringer Ingelheim (sold as combination products like Combivent)
- Teva Pharmaceuticals
- Mylan
Regulatory approvals:
Ipratropium bromide’s patent expired in the early 2000s, opening the market for generics. Current approvals cover inhalation solutions and aerosols for COPD and asthma.
Market Drivers
-
Rising COPD prevalence:
According to WHO, COPD affected 200 million worldwide in 2019. The growing aging population further increases disease burden. -
Prescription patterns:
Physicians favor inhaled therapies like ipratropium bromide due to fewer systemic side effects compared to oral medications. -
Therapeutic positioning:
Ipratropium bromide is often used as a first-line or maintenance therapy in non-acute settings, complementing or replacing older treatments.
Market Challenges
-
Generic competition:
Price erosion limits profit margins. The availability of cost-effective generics depresses sales prices. -
Formulation limitations:
Less effective than newer long-acting anticholinergics for some patients, reducing volume growth potential. -
Regulatory shifts:
New guidelines favor long-acting agents over short-acting formulations, impacting sales.
Sales Projections (2023-2028)
| Year | Projected Market Size (USD Billion) | CAGR | Notes |
|---|---|---|---|
| 2023 | ₹6.5 – 7.2 | 3% – 4% | Continued generic sales, slow market growth |
| 2024 | 7.2 – 7.5 | 2.8% | Slight increase due to aging populations |
| 2025 | 7.5 – 8.0 | 4% | Uptake in emerging markets |
| 2026 | 8.0 – 8.6 | 4.2% | Increased diagnosis and prescription adherence |
| 2027 | 8.6 – 9.2 | 4.3% | Growing demand for inhaled therapies in Asia-Pacific |
| 2028 | 9.2 | 3.5% | Maturation of market with stabilized growth |
(Note: Figures are estimates based on industry reports from IQVIA, MarketsandMarkets, and GlobalData.)
Sales volume projections:
In terms of units, inhalers containing ipratropium bromide are forecasted to grow at approximately 3-4% annually. The total global sales volume is expected to reach around 150-170 million inhalers by 2028.
Emerging market outlook:
A significant growth contributor will be Asian markets with expanding healthcare infrastructure and a rise in respiratory disease diagnosis. India and China combined are projected to account for over 30% of global sales by 2028.
Strategic Outlook
-
Formulation innovation:
Focusing on combination therapies (e.g., ipratropium with albuterol) could preserve market share amid competition. -
Pricing strategies:
Aligning with generics pricing dynamics is necessary to maintain volume sales. -
Regulatory navigation:
Monitoring evolving guidelines favoring long-acting agents could limit future sales unless reformulated or repositioned.
Key Takeaways
- Ipratropium bromide’s market size in 2022 was approximately $4–5 billion globally for inhaled anticholinergics.
- The market exhibits slow to moderate growth, approximately 3-4% annually, driven mainly by emerging markets.
- Competition from generics and newer long-acting bronchodilators constrains rapid growth.
- Asia-Pacific markets are expected to catalyze future sales increases due to rising COPD prevalence.
- Innovation in formulation and strategic positioning will influence long-term market share.
FAQs
1. What is the primary therapeutic use of ipratropium bromide?
It is primarily used for relieving symptoms of COPD and asthma by providing bronchodilation through inhalation.
2. How does the market for ipratropium bromide compare to newer long-acting bronchodilators?
Long-acting agents such as tiotropium and umeclidinium are increasingly preferred for maintenance therapy, reducing the growth of short-acting agents like ipratropium.
3. What factors most influence sales declines?
Patent expirations, price competition from generics, and shifting clinical guidelines favoring long-acting agents.
4. Which regions present the most growth opportunity?
Asia-Pacific, especially India and China, due to demographics and increasing healthcare access.
5. How might future innovations affect the market?
Combination inhalers, dry powder formulations, and personalized inhaler devices could sustain or boost sales.
References
[1] IQVIA, Market Intelligence Reports, 2022.
[2] MarketsandMarkets, "Respiratory Drugs Market," 2021.
[3] GlobalData, "Inhalation Therapy Market Analysis," 2023.
[4] WHO, "Chronic Obstructive Pulmonary Disease Fact Sheet," 2021.
More… ↓
