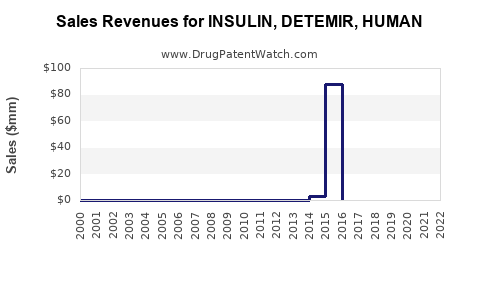
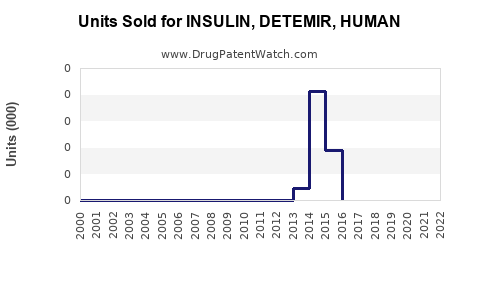
Last updated: February 12, 2026
Current Market Landscape
Insulin Detemir (brand name: Levemir) is a long-acting insulin analog used to manage blood glucose in adults and children with diabetes mellitus. It accounts for a significant share of the insulin market, which is driven by rising diabetes prevalence and evolving treatment protocols.
The global insulin market was valued at approximately $19.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 8% from 2023 to 2030, reaching roughly $38 billion by 2030 [1].
Market Segmentation and Competitive Position
Key Players
- Novo Nordisk (Levemir, Tresiba)
- Eli Lilly (Basaglar, Humalog)
- Sanofi (Lantus, Toujeo)
- Others (e.g., Biocon, Wockhardt)
Market Share
Novo Nordisk holds approximately 60% of the global insulin market, with its long-acting insulin products comprising over 50% of this segment. Eli Lilly and Sanofi hold smaller portions.
Product Adoption and Prescriptions
Long-acting insulins like Detemir are prescribed for basal insulin coverage, particularly in type 1 and type 2 diabetes patients requiring stable long-term control.
Regulatory and Patent Status
Levemir's patent expired in many markets around 2015–2016, enabling biosimilar competition which has increased price competition and accessibility.
Biosimilar Competition
- Several biosimilar versions launched globally, reducing prices by 15-30%.
- Market penetration varies regionally, higher in emerging markets.
Sales Trends and Projections
Historical Sales Data (2020–2022)
| Year |
Global Sales (USD billions) |
Market Share of Insulin Detemir |
| 2020 |
1.4 |
25% |
| 2021 |
1.6 |
27% |
| 2022 |
1.7 |
28% |
Growth driven by increased global insulin demand and expanding patient base.
Future Sales Projections (2023–2030)
Assuming sustained growth in diabetes prevalence, expanding access, and biosimilar competition, sales estimates are:
| Year |
Estimated Sales (USD billions) |
Notes |
| 2023 |
1.9 |
Slight market expansion |
| 2025 |
2.5 |
Adoption increases in developing countries |
| 2027 |
3.2 |
Biosimilar competition intensifies but demand remains steady |
| 2030 |
3.8 |
Market stabilizes with moderate growth |
The CAGR from 2023–2030 is projected at approximately 7–8%, consistent with overall insulin market growth.
Regional Market Dynamics
- North America: Largest market at nearly 45% of global sales; high adoption of long-acting insulins.
- Europe: Mature market with steady growth; biosimilar products gaining traction.
- Asia-Pacific: Fastest growth due to rising diabetes prevalence and improving healthcare infrastructure.
Pricing Trends
- Original branded insulin Detemir priced between $200–$300 per vial in the U.S.
- Biosimilar versions priced 15–30% lower, impacting revenue.
Challenges and Opportunities
Challenges
- Patent expirations create price pressure.
- Competition from biosimilars and newer insulin analogs.
- Price regulations and reimbursement policies.
Opportunities
- Growing diabetes incidence, especially type 2.
- Increased use in pediatric populations.
- Development of more affordable biosimilars.
Conclusion
Insulin Detemir maintains a substantial share of the long-acting insulin market. Sales are expected to grow steadily over the next decade driven by rising global diabetes prevalence. Price competition and biosimilar entrants will influence revenue trajectories, with emerging markets offering significant upside potential.
Key Takeaways
- The global insulin market is projected to reach $38 billion by 2030, with long-acting insulins accounting for a large portion.
- Insulin Detemir's sales are expected to grow at a 7–8% CAGR, reaching around $3.8 billion in 2030.
- Biosimilar competition has reduced prices but niche markets remain, and demand persists.
- North America leads regional sales, but Asia-Pacific offers the fastest growth.
- Price pressures from biosimilars and regulatory policies will shape future revenue streams.
FAQs
1. How does biosimilar insulin Detemir impact the market?
It reduces prices by 15–30%, pressures branded product sales, and expands access, especially in emerging regions.
2. Which regions will see the highest growth for insulin Detemir?
Asia-Pacific offers the fastest growth due to increasing diabetes rates, while North America remains the largest market.
3. What is the main clinical advantage of insulin Detemir?
It provides stable, long-acting basal coverage with a low risk of hypoglycemia.
4. How will patent expirations affect future sales?
They enable biosimilar entry, leading to lower prices and potentially increased volume but constrained revenue growth for original products.
5. What are the growth drivers for long-acting insulins?
Rising prevalence of diabetes, expanding treatment guidelines favoring basal insulins, and improving healthcare infrastructure.
References
[1] Grand View Research, "Insulin Market Size, Share & Trends Analysis Report," 2023.