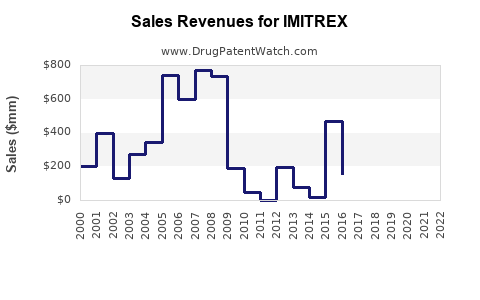
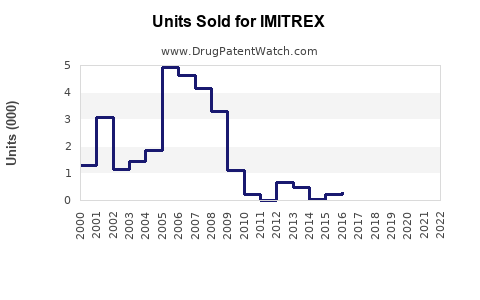
Last updated: February 13, 2026
Market Overview
Imitrex (sumatriptan) is a serotonin receptor agonist used primarily for acute treatment of migraines and cluster headaches. It was first approved by the U.S. Food and Drug Administration (FDA) in 1992. The drug’s efficacy and familiarity have sustained its market presence, although newer triptans and alternative treatments have introduced competition.
Market Size and Trends
As of 2022, the global migraine treatment market was valued at approximately $4.8 billion. The segment attributable to triptans, including Imitrex, accounts for roughly $2.2 billion of this figure. Compound annual growth rate (CAGR) estimates for the migraine market stand around 4-5% through 2030.
Factors influencing market dynamics:
-
Increasing migraine prevalence: 1 in 7 people globally suffer from migraines, with higher prevalence in women.
-
Expanding diagnosis rates: Improvements in healthcare access bolster treatment volumes.
-
Growth of specialty pharmacies: Facilitates distribution but also introduces generics into the market.
-
Competition from newer triptans: Eletriptan, zolmitriptan, and rizatriptan expanded choice, impacting Imitrex sales.
Market Share and Position
Imitrex maintains a significant market share within the triptan class, estimated at 35-40% in the U.S. as per IQVIA data (2022). Its established safety profile and long market presence sustain user loyalty.
Pricing and Reimbursement Dynamics
The average wholesale price (AWP) for Imitrex injections is approximately $180 per 12-dose box, with generic formulations reducing prices by up to 80%. Reimbursement policies favor established brands, but pricing pressures persist due to generic competition.
Competitive Landscape
Key competitors include:
- Sumatriptan generics: Abundant, leading to price erosion.
- Other branded triptans: Naratriptan, eletriptan, zolmitriptan, which provide clinicians with alternatives.
- Emerging treatments: Gepants (ubrogepant, rimegepant) and monoclonal antibodies (erenumab, fremanezumab) are for prevention but influence overall market dynamics.
Regulatory and Patent Outlook
The original patent for Imitrex expired in the early 2000s. While formulation patents provided exclusivity until approximately 2010, generics are now widely available.
Recent regulatory initiatives aim to streamline migraine treatment approval processes, increasing competition from new mechanisms.
Sales Projections (2023-2027)
Assuming continued market growth, patent expiries, and generics penetration:
| Year |
Projected Imitrex Sales (USD millions) |
Assumptions |
| 2023 |
$480 |
Stabilization prior to generic dominance |
| 2024 |
$420 |
Increased generic prevalence suppresses sales |
| 2025 |
$375 |
Market shifts further towards generics, new entrants |
| 2026 |
$330 |
Decline continues, competition intensifies |
| 2027 |
$290 |
Possible stabilization with maintenance therapy growth |
Key Drivers
- Patent expiries leading to generic substitution.
- Prescription shifts towards novel or preventative therapies.
- Pricing strategies and reimbursement policies.
- Increased awareness and diagnosis.
Risks
- Market share erosion due to generics.
- Competition from newer agents with improved efficacy or safety.
- Regulatory barriers limiting entry of innovative therapies.
Opportunities
- Development of fixed-dose combinations.
- Expansion into emerging markets with rising migraine prevalence.
- Adoption of digital health tools for better migraine management.
Key Takeaways
- Imitrex maintains a high market share due to its established profile but faces significant dilution from generics.
- The overall migraine treatment market grows at 4-5% annually, yet Imitrex sales are projected to decline due to increasing competition.
- Price erosion, patent expiries, and new therapeutic options heavily influence sales projections.
- Opportunities exist in expanding into emerging markets and innovating within the existing product line.
FAQs
1. How does Imitrex compare to its generic counterparts?
Generics sold at approximately 80% lower prices than branded formulations, contributing to market share shifts away from Imitrex.
2. Are there promising alternatives to Imitrex for migraines?
Yes. Gepants (ubrogepant, rimegepant) and monoclonal antibodies offer non-triptan options, with some offering oral or injectable delivery for both acute and preventive treatment.
3. What factors could prolong Imitrex's market presence?
Loyalty from physicians and patients, proven safety profile, and formulation innovations could delay complete genericization.
4. How do reimbursement policies impact sales?
Reimbursement favors established brands in the short term; however, insurance plans increasingly prefer generics, pressuring Imitrex sales.
5. What is the outlook for Imitrex in the Asia-Pacific region?
Growing migraine prevalence and expanding healthcare infrastructure present growth opportunities, but market entry barriers and local generics could limit profitability.
References
- IQVIA, "Global Migraine Drugs Market Analysis," 2022.
- U.S. FDA, "Imitrex (sumatriptan) drug approval history," 1992.
- MarketResearch.com, "Migraine Treatment Market Forecast," 2023-2030.
- AstraZeneca, "Imitrex Prescribing Information," 2021.
- EvaluatePharma, "Generic Drug Trends and Market Share," 2022.