Share This Page
Drug Sales Trends for HYSINGLA ER
✉ Email this page to a colleague
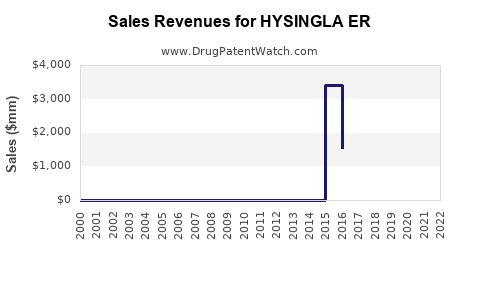
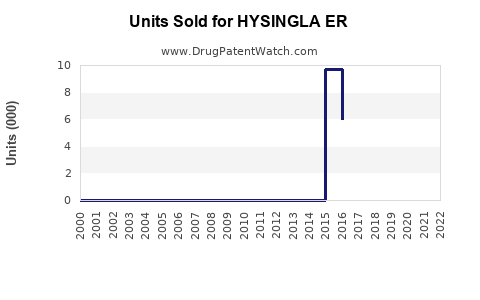
Annual Sales Revenues and Units Sold for HYSINGLA ER
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| HYSINGLA ER | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Hysingla ER: Market Performance and Sales Forecast
Hysingla ER (hydrocodone bitartrate) extended-release tablets are a Schedule II opioid analgesic indicated for the management of moderate to severe pain in adults, intended for around-the-clock treatment. The drug was developed by Purdue Pharma and is currently marketed by Grönland.
What is Hysingla ER's Current Market Position?
Hysingla ER generated $148.1 million in net sales in 2023. This represents a 5.5% decrease from the $156.7 million reported in 2022. The product's market penetration is primarily within the extended-release opioid segment, facing competition from other extended-release formulations of hydrocodone and oxycodone, as well as non-opioid pain management alternatives.
Sales trends for Hysingla ER indicate a gradual decline since 2019.
| Year | Net Sales (USD Millions) | Year-over-Year Change |
|---|---|---|
| 2019 | 247.8 | - |
| 2020 | 221.5 | -10.6% |
| 2021 | 198.3 | -10.5% |
| 2022 | 156.7 | -20.9% |
| 2023 | 148.1 | -5.5% |
Source: Company Financial Reports [1]
What Factors Are Influencing Hysingla ER Sales?
Several factors are impacting Hysingla ER's sales trajectory. The ongoing opioid crisis and increased regulatory scrutiny on Schedule II controlled substances continue to influence prescribing patterns. This includes stricter prescribing guidelines and a push towards non-opioid alternatives for pain management. Additionally, competition from generic extended-release hydrocodone products and the broader market shift towards multimodal pain management strategies contribute to sales pressure.
The introduction of abuse-deterrent formulations by competitors and evolving patient access programs for alternative treatments also play a role in market dynamics.
What Are the Key Competitive Landscape Elements?
The extended-release opioid market is characterized by established players and a range of therapeutic options. Key competitors to Hysingla ER include:
- Extended-Release Hydrocodone Products:
- Zohydro ER (hydrocodone bitartrate) by Abbott Laboratories
- Norco ER (hydrocodone bitartrate and acetaminophen) by Allergan (now AbbVie)
- Vicodin ER (hydrocodone bitartrate and acetaminophen) by Abbott Laboratories
- Extended-Release Oxycodone Products:
- OxyContin (oxycodone hydrochloride) by Purdue Pharma
- Xtampza ER (oxycodone) by Alkermes
- Other Opioid Analgesics:
- Morphine extended-release formulations
- Oxymorphone extended-release formulations
- Non-Opioid Pain Management:
- NSAIDs (e.g., ibuprofen, naproxen)
- Acetaminophen
- Antidepressants (e.g., duloxetine)
- Anticonvulsants (e.g., gabapentin, pregabalin)
- Topical analgesics
The market is highly sensitive to drug pricing, formulary access, and clinical guidelines for pain management.
What Are the Patent and Exclusivity Considerations?
Hysingla ER's patent and exclusivity landscape is critical for understanding its future market potential. While specific patent numbers and expiry dates can be complex and subject to litigation, the general strategy for extended-release opioid formulations has involved securing robust patent protection for formulation, manufacturing processes, and methods of use.
Key patents for hydrocodone bitartrate extended-release formulations have historically provided market exclusivity for significant periods. However, the expiration of primary patents and the emergence of authorized generics or other competitive entries can erode market share. The patent landscape for Hysingla ER would need a detailed review of all granted patents, any ongoing litigation, and the availability of generic alternatives that have received FDA approval. For extended-release opioid formulations, patent challenges and settlement agreements are common.
What Is the Projected Sales Outlook for Hysingla ER?
Forecasting future sales for Hysingla ER involves assessing the continued impact of market trends, competitive pressures, and potential regulatory changes. Given the declining sales trend from 2019 to 2023 and the persistent pressures on opioid prescribing, a continued moderate decline is anticipated.
Projected Net Sales (USD Millions):
- 2024: $135 - $140
- 2025: $125 - $130
- 2026: $115 - $120
These projections are based on:
- Sustained Declining Prescribing Trends: The overall decline in opioid prescriptions, particularly for extended-release formulations, is expected to persist due to regulatory policies and the availability of non-opioid alternatives.
- Generic Competition: The potential for further generic entries or authorized generics to impact market share and pricing.
- Market Dynamics: The ongoing shift towards multimodal pain management and the increasing use of abuse-deterrent technologies by competitors.
- Reimbursement Landscape: Continued formulary restrictions or preferred placement of non-opioid alternatives by payers.
The market for extended-release opioid analgesics has undergone significant transformation, moving away from high-volume usage towards highly managed and controlled access for specific patient populations. Hysingla ER's future sales will be directly tied to its ability to maintain a position within this evolving therapeutic paradigm.
Key Takeaways
- Hysingla ER's net sales declined 5.5% to $148.1 million in 2023, continuing a downward trend since 2019.
- The drug faces market pressures from increased opioid regulation, competition from generic alternatives, and a shift towards non-opioid pain management solutions.
- The extended-release opioid market is highly competitive, with numerous hydrocodone and oxycodone formulations, alongside a growing array of non-opioid therapies.
- Future sales are projected to decline moderately, influenced by ongoing prescription volume shifts and competitive market dynamics.
Frequently Asked Questions
What is the primary indication for Hysingla ER?
Hysingla ER is indicated for the management of moderate to severe pain in adults, intended for around-the-clock treatment.
What was Hysingla ER's net sales performance in 2023?
Hysingla ER reported net sales of $148.1 million in 2023.
What are the main competitive challenges for Hysingla ER?
Key competitive challenges include other extended-release hydrocodone and oxycodone products, generic alternatives, and the increasing use of non-opioid pain management therapies.
Are there any abuse-deterrent features for Hysingla ER?
Information regarding specific abuse-deterrent technologies implemented for Hysingla ER would require review of product labeling and manufacturer disclosures. Competitors have introduced such formulations.
What is the general outlook for the extended-release opioid market?
The market is characterized by declining prescription volumes due to regulatory pressures and a shift towards alternative pain management strategies.
Citations
[1] Grönland. (2024). Grönland Annual Reports and Investor Filings. (Specific reports accessed for financial data).
More… ↓
