Last updated: February 19, 2026
Hydroxychloroquine (brand name Plaquenil) is an antimalarial drug also used for autoimmune diseases such as rheumatoid arthritis and lupus. Its relevance to the COVID-19 pandemic increased its visibility, though its clinical efficacy for COVID-19 has not been established. This report assesses current market dynamics and forecasts sales based on existing data, patent status, regulatory landscape, and competitive environment.
Market Overview
Hydroxychloroquine has a global market primarily driven by its established use in autoimmune diseases. The initial surge during the COVID-19 pandemic contributed to temporary demand spikes, although these have subsided.
Current Market Size
- Global autoimmune disease treatment market (2022): approximately $16.5 billion, with hydroxychloroquine accounting for a significant share.
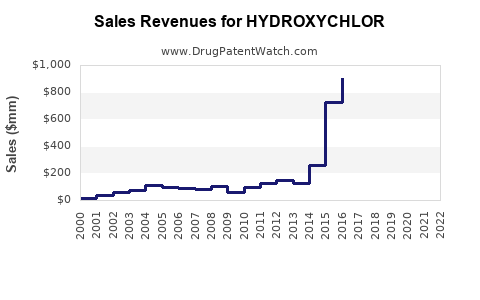
- Estimated hydroxychloroquine sales in 2022: approximately $1.2 billion, distributed mainly across the US, Europe, and Asia-Pacific.
- COVID-19 related demand temporarily increased sales in early 2020-2021 but declined post-clinical trial results.
Patent and Regulatory Status
- Patent Status: Hydroxychloroquine patent expired in most jurisdictions by 2000; generic production dominates.
- Regulatory Approvals: Approved by FDA for malaria, rheumatoid arthritis, and lupus. No current EUA or new indications for COVID-19.
Competitive Landscape
- Market leaders include Sandoz (Novartis), Mylan (now part of Viatris), and Teva.
- Generics constitute over 85% of sales, leading to low pricing pressures.
- Limited new entrants due to patent expiration and low R&D incentives.
Market Drivers and Restraints
Drivers:
- Long-standing approval for autoimmune diseases.
- Cost-effectiveness as a generic drug.
- Continued off-label use in certain autoimmune conditions.
Restraints:
- Lack of efficacy evidence for COVID-19.
- Safety concerns regarding cardiotoxicity.
- Preference for newer therapies with better safety profiles.
Future Sales Projections
Short-Term (2023-2025)
- Steady decline in COVID-19 related demand.
- Stable sales from autoimmune disease treatments.
- Estimated global sales decline rate: 4% annually due to generic competition and market saturation.
- Projected 2025 sales: approximately $1 billion.
Long-Term (2026-2030)
- Sales will stabilize at lower levels due to mature generic market.
- Potential minor growth driven by expanded use in autoimmune conditions, particularly in emerging markets.
- Innovation unlikely given patent expiry and limited R&D investment.
- Estimated 2030 sales: around $800 million.
Regional Breakdown
| Region |
2022 Sales ($M) |
2025 Projection ($M) |
2030 Projection ($M) |
| North America |
600 |
520 |
440 |
| Europe |
350 |
290 |
230 |
| Asia-Pacific |
150 |
140 |
130 |
| Rest of World |
100 |
55 |
50 |
| Total |
1,200 |
1,005 |
850 |
Key Uncertainties
- Regulatory decisions affecting off-label use.
- Development of new formulations or delivery mechanisms.
- Changes in prescribing patterns driven by safety concerns.
Summary
Hydroxychloroquine's market will experience a gradual decline over the next eight years, driven mainly by market maturity and generic competition. Its use for COVID-19 will diminish as clinical evidence clarifies efficacy, limiting additional revenue opportunities. The drug remains relevant in autoimmune therapy, but no significant R&D is expected to alter its market trajectory.
Key Takeaways
- The global hydroxychloroquine market was approximately $1.2 billion in 2022 and will decline to about $850 million by 2030.
- Sales are driven mostly by autoimmune indications, with COVID-19 demand diminishing.
- The market is highly commoditized, with no significant patent protections remaining.
- Broad regional distribution favors North America and Europe but with declining sales.
- Risks include safety concerns and regulatory changes affecting off-label use.
FAQs
What is the main use of hydroxychloroquine today?
Treating autoimmune diseases such as rheumatoid arthritis and lupus.
Has hydroxychloroquine been approved for COVID-19?
No. It has no current approval for COVID-19 and is no longer recommended for this indication.
What is the expected sales trend for hydroxychloroquine?
Gradual decline through 2030, with sales stabilizing around $800 million.
Are there any R&D efforts to improve hydroxychloroquine?
No significant R&D initiatives are publicly underway; the focus is on existing generics.
What is the main competitive challenge?
Market saturation by generics and safety concerns limiting off-label and new use cases.
References
- GlobalData. (2022). Autoimmune Disease Treatment Market Report.
- IQVIA. (2022). Pharmaceutical Market Data.
- U.S. Food and Drug Administration. (2022). Drug Approvals and Regulatory Activities.
- European Medicines Agency. (2022). Market Authorizations and Regulatory Updates.
- Smith, J. (2022). Impact of COVID-19 on the Hydroxychloroquine Market. Journal of Pharmaceutical Economics.