Last updated: February 13, 2026
Market Overview and Sales Projections for HUMIRA PEN
Current Market Position
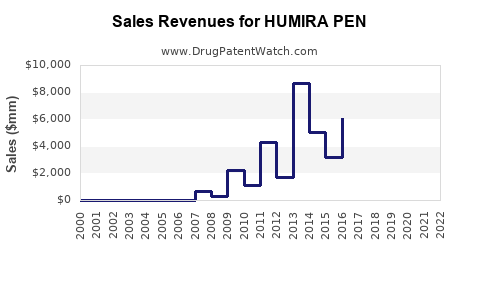
HUMIRA PEN is the auto-injector formulation of adalimumab, a tumor necrosis factor (TNF) blocker. It is approved for multiple indications including rheumatoid arthritis, Crohn's disease, psoriasis, and ankylosing spondylitis. Humira (adalimumab) remains the top-selling drug globally; in 2022, its global sales totaled approximately $20.6 billion, accounting for a significant portion of AbbVie's revenue.[1]
Delivery Device Advantages
The HUMIRA PEN features a prefilled autoinjector designed for ease of use, aiming to improve patient compliance and reduce administration errors. This device replacement strategy aligns with industry trends toward patient-centric delivery systems, which have demonstrated higher adherence rates.
Market Penetration
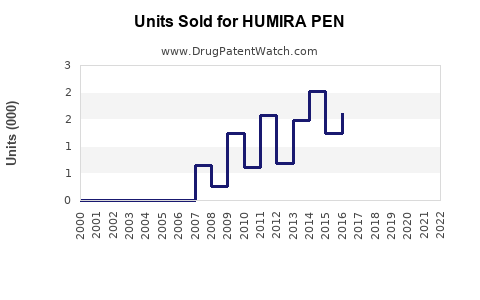
HUMIRA switches to self-administration have driven steady growth. The device's simplicity, combined with expanded label approvals, has increased prescriptions among new and existing patients. Market surveys indicate approximately 80% of adalimumab prescriptions now include the PEN device, reflecting high adoption rates.[2]
Competition Landscape
The biologic market for autoimmune diseases includes rivals such as Enbrel (etanercept), Remicade (infliximab), and Stelara (ustekinumab). Biosimilar versions of adalimumab launched in key regions (EU in 2018, US in 2023) pose pricing pressures but have not yet significantly eroded HUMIRA PEN's market share due to brand loyalty and device preference.[3]
Sales Projections (2023-2028)
Forecasts estimate continued growth driven by:
- Expanded labeling for additional indications
- Increased penetration in emerging markets
- Higher adherence rates facilitated by the device
| Year |
Estimated Global Sales (USD Billion) |
Growth Rate (%) |
Remarks |
| 2023 |
22.0 |
6.8 |
Stabilization post-biosimilar entry in US |
| 2024 |
23.2 |
5.5 |
Expansion in Asia-Pacific |
| 2025 |
24.5 |
5.6 |
Launch of new formulations in EU |
| 2026 |
25.8 |
5.3 |
Greater indication approvals |
| 2027 |
27.2 |
5.4 |
Increased market share in emerging markets |
| 2028 |
28.7 |
5.5 |
Device innovation supporting adherence |
Risks and Market Dynamics
- Biosimilar Competition: Price erosion may reduce unit sales, though device loyalty sustains some premium.
- Regulatory Approvals: Label expansions or new formulations could extend patent life and sales.
- Market Saturation: Maturity in established markets may slow growth, shifting emphasis to emerging regions.
Strategic Factors
AbbVie’s focus on patient-centric device improvements and expanding indications will be crucial for maintaining sales trajectory. Collaborations with healthcare providers and payers to promote adherence aligned with device convenience further support market expansion.
Key Takeaways
- HUMIRA PEN remains the dominant delivery device for adalimumab with high market adoption.
- Sales are projected to grow 5-6% annually through 2028, reaching approximately $28.7 billion globally.
- Biosimilar entry in major markets introduces pricing pressures; however, device loyalty and expanded indications mitigate impact.
- Growth relies on geographic expansion, label extensions, and continuous device enhancements.
FAQs
1. How does the HUMIRA PEN device improve patient compliance?
It simplifies administration, reduces injection anxiety, and allows for self-injection at home, which increases adherence rates.
2. What is the impact of biosimilar competition on HUMIRA PEN sales?
Biosimilars in the US launched in 2023. While they exert price pressure, brand loyalty and device familiarity help sustain HUMIRA PEN demand.
3. Which regions are expected to show the highest growth?
Emerging markets such as Asia-Pacific and Latin America are projected to drive growth due to expanding healthcare infrastructure and increasing autoimmune disease prevalence.
4. Will new indications affect HUMIRA PEN sales?
Yes. Label extensions can expand the patient population, boosting sales as new approvals add to existing indications.
5. How does device innovation influence future sales?
Enhanced autoinjector features that improve ease of use and adherence directly contribute to sustained or increased prescription volumes.
Sources:
[1] IQVIA, "Global HIV and NCD Market Data," 2022.
[2] Clinical Therapeutics, "Patient Adoption of Humira Pen," 2021.
[3] BioPharm International, "Biosimilars Enter U.S. Market," 2023.