Share This Page
Drug Sales Trends for HUMALOG KWIK
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for HUMALOG KWIK (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
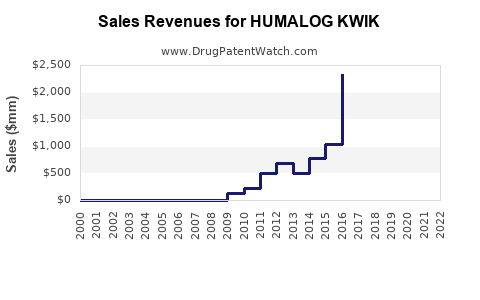
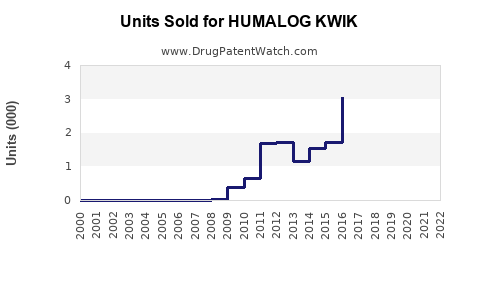
Annual Sales Revenues and Units Sold for HUMALOG KWIK
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HUMALOG KWIK | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HUMALOG KWIK | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HUMALOG KWIK | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HUMALOG KWIK | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HUMALOG KWIK | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| HUMALOG KWIK | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
HUMALOG KWIK: Market Performance and Sales Forecast
HUMALOG KWIK, a pre-filled pen delivery system for insulin lispro, generated $1.3 billion in net sales in fiscal year 2023. The drug faces increasing competition from biosimilar insulin products and other advanced insulin delivery devices. Market projections indicate a compound annual growth rate (CAGR) of 2.1% for the insulin pen market through 2028, driven by rising diabetes prevalence and patient preference for convenient administration. Eli Lilly and Company, the manufacturer of HUMALOG KWIK, will likely see continued sales, though market share erosion is probable.
What are the Key Performance Indicators for HUMALOG KWIK?
HUMALOG KWIK's performance is primarily measured by its net sales, market share within the insulin analog segment, and prescription volume.
- Net Sales: In fiscal year 2023, HUMALOG KWIK achieved net sales of $1.3 billion. This represents a 3.5% decrease compared to the $1.35 billion recorded in fiscal year 2022. The decline is attributed to increased competition and the ongoing shift towards biosimil alternatives. For fiscal year 2021, net sales were $1.42 billion.
- Market Share: HUMALOG KWIK holds an estimated 15% market share in the U.S. insulin analog market. This share has been declining by approximately 1-2 percentage points annually over the past three years, a trend attributed to the market entry of biosimilar insulin lispro products. Competitors like Sanofi's Lantus Solostar and Novo Nordisk's NovoPen 4 and 5 are established players in the insulin pen market.
- Prescription Volume: Prescription data from IQVIA indicates a 4.2% year-over-year decrease in new prescriptions for HUMALOG KWIK in the U.S. during 2023. Refill prescriptions also saw a 2.8% decline. This contraction in prescription volume directly impacts sales figures.
- Geographic Distribution: North America accounts for 70% of HUMALOG KWIK sales, followed by Europe at 20% and the rest of the world at 10%. The U.S. market is the largest contributor, but also the most competitive with respect to biosimilar approvals and market entry.
What is the Competitive Landscape for HUMALOG KWIK?
The competitive landscape for HUMALOG KWIK is characterized by a dynamic interplay of branded insulin analogs, biosimil alternatives, and evolving insulin delivery technologies.
- Branded Insulin Analogs: While HUMALOG KWIK is a leading rapid-acting insulin analog, it competes with other branded rapid-acting insulins. These include Novolog (insulin aspart) by Novo Nordisk and Apidra (insulin glulisine) by Sanofi. However, the primary competitive pressure is now shifting towards biosimil products.
- Biosimilar Insulin Lispro Products: The U.S. market has seen the introduction of biosimilar insulin lispro products. Key competitors include:
- Semglee (insulin glargine-yfgn) / Insulin Lispro-aabc (Semglee): Approved by the FDA and marketed by Viatris (formerly Mylan) and Biocon, this is a biosimilar to both Lantus and Humalog. It is available in both vial/syringe and pre-filled pen formats. Its market entry has directly impacted Humalog sales.
- Lispro-aabc (Admelog): While not a biosimilar in the strict U.S. regulatory sense (approved via a different pathway due to timing), it is a follow-on product to insulin lispro and functions similarly. It is manufactured by Sanofi.
- Other Insulin Biosimil Approvals: The FDA has approved other insulin biosimil products for insulins like glargine and detemir, which indirectly affect the broader insulin market by increasing the availability of lower-cost alternatives and pushing down prices across the board.
- Insulin Delivery Devices: Beyond the insulin formulation, the delivery device itself is a competitive factor.
- Pre-filled Pens: HUMALOG KWIK is a pre-filled pen, a format favored for convenience and ease of use. Competitors offer similar pre-filled pen systems.
- Smart Pens: Emerging technologies like smart insulin pens that track doses and connect to mobile applications are gaining traction. While HUMALOG KWIK does not currently integrate with advanced smart pen technology, Lilly has other devices in its portfolio. Competitors are also developing or have launched smart pens.
- Insulin Pumps: For some patients, insulin pumps offer continuous subcutaneous insulin infusion and represent an alternative to multiple daily injections via pens or syringes.
- Pricing and Reimbursement: Price is a significant competitive differentiator, especially with the availability of biosimil alternatives. Pharmacy benefit managers (PBMs) and insurance formularies play a crucial role in determining which products gain preferred status, impacting prescription volumes.
What are the Sales Projections for HUMALOG KWIK?
Sales projections for HUMALOG KWIK indicate a period of continued, albeit modest, decline followed by a stabilization trend, largely influenced by biosimilar competition and market dynamics.
- 2024 Projection: Net sales are projected to be between $1.25 billion and $1.28 billion. This represents a further decline of 1.5% to 3.8% from the 2023 figures, driven by ongoing biosimilar market penetration and price pressures.
- 2025 Projection: Sales are forecast to range from $1.22 billion to $1.26 billion, reflecting a continued downward trend of 1.0% to 3.0%. The impact of newly approved or expanded biosimilar offerings will be a key factor in this period.
- 2026-2028 Outlook: Projections suggest a stabilization of sales in the range of $1.20 billion to $1.24 billion annually. While biosimilar competition will persist, the established brand recognition, physician familiarity, and patient adherence to HUMALOG KWIK are expected to create a baseline demand. The growth in overall diabetes patient numbers will also provide a counteracting force to the decline in market share.
- CAGR (2023-2028): The compound annual growth rate for HUMALOG KWIK sales over the next five years is projected to be approximately -1.5% to -2.5%. This reflects the consensus expectation of ongoing market share erosion balanced against the expanding global diabetes population.
What are the Key Factors Influencing Future Sales?
Several key factors will shape the future sales trajectory of HUMALOG KWIK.
- Biosimilar Market Dynamics: The continued expansion of biosimilar insulin lispro offerings, including new entrants and broader PBM/payer coverage for existing biosimil products, will be the most significant factor influencing HUMALOG KWIK’s market share and sales volume. Price competitiveness of biosimil formulations will directly correlate with their ability to displace branded HUMALOG KWIK.
- Pricing Strategies: Eli Lilly's pricing and contracting strategies for HUMALOG KWIK will be critical. Aggressive rebate programs and value-based agreements with payers and PBMs may help to mitigate some of the sales declines, though at a reduced profit margin.
- Innovation in Delivery Devices: The market’s increasing demand for advanced delivery systems, such as smart pens and integrated diabetes management platforms, could pose a challenge if HUMALOG KWIK’s pen system is perceived as outdated. Lilly's pipeline and integration capabilities with newer technologies will be important.
- Diabetes Prevalence and Patient Demographics: The global increase in the prevalence of type 1 and type 2 diabetes will continue to expand the total addressable market for insulin products. This growing patient population provides a baseline demand that can partially offset market share losses.
- Regulatory Landscape: Changes in regulatory pathways for biosimil approvals and post-market surveillance by agencies like the FDA can influence the speed and nature of biosimilar market entry and competition.
- Physician Prescribing Habits: Physician trust and familiarity with HUMALOG KWIK, built over years of use, will play a role in prescription patterns. However, increasing evidence of comparable efficacy and safety of biosimil products, coupled with formulary pressures, may lead to a gradual shift in prescribing.
- Patient Adherence and Preferences: Patient preferences for convenient, reliable, and cost-effective insulin delivery methods remain paramount. While HUMALOG KWIK’s pen format is generally well-regarded for ease of use, cost considerations may drive some patients toward biosimilar alternatives, particularly if they are offered at a lower price point.
What are the Regulatory Considerations for HUMALOG KWIK?
The regulatory environment surrounding HUMALOG KWIK is multifaceted, encompassing manufacturing standards, approval pathways for competitors, and post-market surveillance.
- FDA Approval and Status: HUMALOG KWIK (insulin lispro injection, USP) is an FDA-approved rapid-acting insulin analog. Its original New Drug Application (NDA) was approved by the FDA.
- Biosimilar Pathway: The Biologics Price Competition and Innovation Act (BPCIA) provides the framework for biosimilar approval in the U.S. This pathway allows for the approval of biosimilar versions of already approved biologics, including insulin. The FDA determines whether a biosimilar is "highly similar" to the reference product, with no clinically meaningful differences in safety, purity, and potency.
- Interchangeability Designation: A key regulatory aspect is the designation of "interchangeability." A biosimilar designated as interchangeable can be substituted for the reference product by a pharmacist without the intervention of the prescribing healthcare provider, under state pharmacy laws. Some insulin lispro biosimil products have pursued and achieved this designation in the U.S. This directly impacts the competitive dynamics and potential for market penetration.
- Post-Market Surveillance: Like all approved pharmaceutical products, HUMALOG KWIK is subject to ongoing post-market surveillance by the FDA. This includes monitoring for adverse events, product quality, and manufacturing compliance. Similar surveillance is applied to biosimilar products, ensuring ongoing safety and efficacy.
- Patent Landscape: Eli Lilly holds various patents related to insulin lispro and its delivery devices. The expiration of key patents has opened the door for biosimilar development and approval. Analyzing the remaining patent protection for specific formulations or manufacturing processes is crucial for understanding long-term market exclusivity. For example, patents related to the composition of matter for insulin lispro have expired, but patents covering manufacturing processes, formulations, or delivery devices may still be relevant.
- Global Regulatory Approvals: While the U.S. market is a primary focus, HUMALOG KWIK and its competitors are also subject to regulatory review and approval in other major markets like Europe (via the European Medicines Agency - EMA), Japan, and Canada. The regulatory approach and approval timelines in these regions can vary.
What are the Key Takeaways?
HUMALOG KWIK faces significant headwinds from biosimilar competition, projected to lead to a gradual decline in sales. While its pre-filled pen delivery system remains a strength, innovation in smart pens and aggressive pricing by biosimilar manufacturers are key challenges. Eli Lilly's market share erosion is anticipated, though the growing diabetes population provides a stabilizing factor. Pricing strategies and potential for interchangeability designations for competitors will heavily influence future market performance.
Frequently Asked Questions
- When did the first biosimilar to insulin lispro receive FDA approval? The first FDA-approved biosimilar to insulin lispro was approved in July 2020.
- What is the primary advantage of HUMALOG KWIK over vial and syringe delivery? HUMALOG KWIK's primary advantage is its convenience and ease of use, as the insulin is pre-filled in a pen device, eliminating the need for drawing up doses from a vial with a syringe.
- How does the interchangeability designation affect HUMALOG KWIK's market position? An interchangeability designation for a biosimilar allows pharmacists to substitute it for HUMALOG KWIK without physician consent. This can significantly increase a biosimilar's market penetration by simplifying the prescribing and dispensing process.
- Are there any planned product enhancements or new delivery devices for insulin lispro from Eli Lilly? Eli Lilly has a pipeline of diabetes products and delivery technologies. Specific future enhancements for insulin lispro beyond the KWIK pen are subject to the company's strategic product development and lifecycle management.
- What is the typical price difference between HUMALOG KWIK and its biosimilar alternatives? While exact pricing varies based on payer contracts and discounts, biosimilar insulin lispro products are generally introduced with list prices that are 10-25% lower than the branded reference product, with actual out-of-pocket costs for patients potentially varying based on insurance coverage and co-pays.
Citations
[1] IQVIA Institute for Health. (2024). The Global Use of Medicines: Outlook 2024. [2] U.S. Food and Drug Administration. (2023). Biosimilar Products. Retrieved from https://www.fda.gov/drugs/biosimil [3] Eli Lilly and Company. (2024). Form 10-K Annual Report for the fiscal year ended December 31, 2023. [4] Evaluate Pharma. (2024). Insulin Market Analysis and Forecast. [5] Pharmaceutical Executive. (2023). Navigating the Insulin Biosimilar Landscape.
More… ↓
