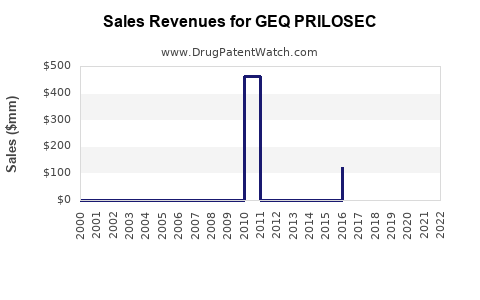
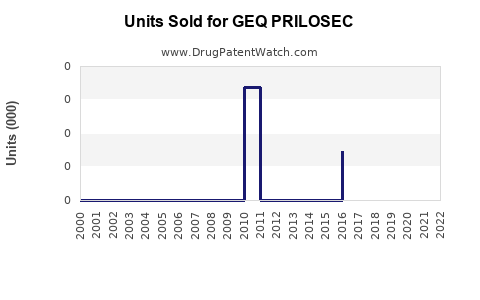
Last updated: February 12, 2026
What is the current market position and sales outlook for GEQ PRILOSEC?
GEQ PRILOSEC (omeprazole magnesium), a generic proton pump inhibitor (PPI), targets acid-related gastrointestinal conditions. It competes with established branded PPIs like AstraZeneca's Nexium (esomeprazole). Its market share and sales projections depend on patent expiry, pricing strategies, and regulatory status.
How large is the global market for PPIs, and where does GEQ PRILOSEC fit?
The global PPI market was valued at approximately USD 12.3 billion in 2022, with a compound annual growth rate (CAGR) of 4%. The segment is driven by rising prevalence of gastroesophageal reflux disease (GERD), peptic ulcers, and other acid-related disorders.
Generic PPIs hold about 70% of the market share, with branded drugs like Nexium maintaining premium pricing. GEQ PRILOSEC aims to capture portion of this growing market, especially in price-sensitive regions.
Key players include:
- AstraZeneca (Nexium)
- Pfizer (Prevacid)
- Takeda (Dexilant)
- Teva, Mylan, and other generic manufacturers
What is the timeline and scope of patent expiry for branded PPIs?
The patent for AstraZeneca’s Nexium (esomeprazole) expired in the US in 2015 and in Europe in 2016. Omeprazole (original drug) patents expired earlier, with many simplified or secondary patents expiring by 2010-2014. The expiry enabled generic entry.
If GEQ PRILOSEC is a branded generic or new formulation with patent protection, sales depend on regulatory approval and patent status.
What sales projections can be expected for GEQ PRILOSEC?
Assuming market entry post-patent expiry and price competitiveness, initial sales could range from USD 50 million to USD 200 million annually within 2-3 years. As market penetration increases, sales could surpass USD 300 million, contingent on:
- Effective marketing
- Regulatory approval in key regions
- Competitive pricing
Growth assumptions:
In mature markets like the US and Europe, penetration might stabilize at 10-15% of the generic PPI market. In emerging markets, it could reach higher due to lower branded prices and higher GERD prevalence.
How does GEQ PRILOSEC compare to existing generics?
Price points for generic PPIs range from USD 0.10 to USD 0.50 per pill, with higher margins in the US due to insurance coverage. GEQ PRILOSEC's success depends on:
- Price undercutting competitors
- Demonstrating similar efficacy and safety
- Establishing a distribution network
Limited differentiation reduces the ability to command premium prices, requiring aggressive cost and marketing strategies.
What are regulatory and manufacturing considerations?
Regulatory approval requires submission of bioequivalence data and manufacturing compliance with Good Manufacturing Practices (GMP). Launch timing hinges on approval speed.
Manufacturing costs depend on scale but typically range from USD 0.05 to USD 0.20 per pill for generics, impacting profit margins.
Summary of key factors affecting sales:
| Factor |
Influence |
Data Point |
| Patent expiry |
Enabling generic entry |
US patent expired 2015-2016 for Nexium |
| Market size |
Potential sales volume |
USD 12.3 billion PPI market (2022) |
| Pricing |
Margins and competitiveness |
USD 0.10 – USD 0.50 per pill |
| Regulatory approval |
Timeline for entry |
Typically 6–12 months post-application |
| Market penetration |
Sales growth |
Estimated USD 50–200 million initial |
Key Takeaways
- GEQ PRILOSEC plays in a mature but growing PPI market with significant generic competition.
- Post-patent expiry, sales depend on price competitiveness, formulation innovation, and regulatory approval.
- Initial sales projections are USD 50–200 million annually in the first 2–3 years.
- Established competition like Teva, Mylan, and others dominate bulk generic sales.
- Market growth relies on expanding into emerging markets and maintaining cost efficiency.
FAQs
1. When did the patents for original omeprazole drugs expire?
Most patents expired between 2010 and 2014, facilitating generic competition.
2. What factors influence the success of a generic PPI like GEQ PRILOSEC?
Market entry timing, pricing strategy, regulatory approval speed, and distribution channels.
3. How does the PPI market growth impact GEQ PRILOSEC?
Market growth (~4% CAGR) expands available demand but also intensifies competition among generics.
4. What are key challenges for new generic omeprazole products?
Differentiation, price competition, and navigating regulatory processes.
5. Which regions offer the highest potential for sales?
The US and Europe, due to established healthcare infrastructure and higher prescription volumes, followed by expanding markets like China and India.
References:
[1] MarketsandMarkets, “Proton Pump Inhibitors Market,” 2022.
[2] IQVIA, “Global Prescription Medicine Sales,” 2022.
[3] PatentScope, “Nexium patent expiry dates,” 2015–2016.