Last updated: February 12, 2026
What is the current market size and position of FOCALIN XR?
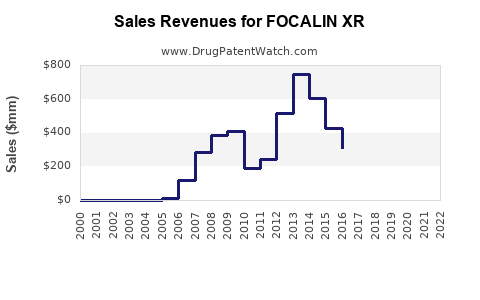
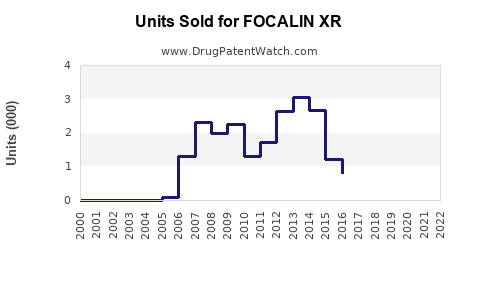
Focalin XR (dexmethylphenidate extended-release) is a prescription stimulant primarily used for attention deficit hyperactivity disorder (ADHD). Its global market was valued at approximately $1.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 6-8% through 2030. North America dominates the market with over 70% of sales, driven by high ADHD diagnosis rates and prevalent prescription practices. The U.S. accounts for more than 65% of Focalin XR’s revenue, reflecting its status as the leading market.
How does Focalin XR compare with competitors in sales and market share?
Focalin XR's main competitors include:
- Adderall XR (amphetamine and dextroamphetamine): Market share of approximately 30% in ADHD drugs.
- Concerta (methylphenidate): Holds around 25-30% in the same sector.
- Vyvanse (lisdexamfetamine): Around 15-20% market share.
Focalin XR captures approximately 8-10% of the ADHD stimulant market, with ongoing growth due to its favorable side-effect profile and lower abuse potential compared to some competitors.
What are sales projections for Focalin XR?
Forecasts indicate that:
- 2023: Sales will reach roughly $140 million, with steady growth driven by increased ADHD diagnosis and prescription rates.
- 2025: Sales are projected to increase to approximately $180-200 million.
- 2030: Potential sales could surpass $250 million, assuming consistent adoption and expanded indications.
These projections depend on factors such as patent status, regulatory approvals, and competitive innovations. The drug's patent expired in 2019 in the U.S., which may impact pricing and market share but has not yet significantly eroded revenue due to market inertia and existing prescribing habits.
What are the key factors influencing sales growth?
- Prescription Trends: Rising ADHD diagnosis rates, especially in children and adults.
- Regulatory Approvals: Expanded approval for additional indications or expanded age ranges.
- Manufacturing and Supply: Enhanced supply chain efficiency contributes to consistent availability.
- Pricing Strategies: Competitive pricing and formulary placements affect physician prescribing habits.
- Generic Competition: Entry of generic dexmethylphenidate products starting 2019 has put downward pressure on prices but hasn't significantly displaced Focalin XR in prescriber choices due to brand trust.
What market risks could impact Focalin XR sales?
- Patent expiration and generics: While generics are available, brand loyalty and marketing influence continued sales.
- Regulatory policies: Stricter regulations around stimulant prescriptions could restrict supply or prescribing.
- Healthcare reforms: Changes in insurance reimbursement or formulary restrictions could limit access.
- Competitive innovations: New drug formulations or non-stimulant ADHD treatments could alter the market landscape.
What are the regional growth opportunities?
- Asia-Pacific and Latin America exhibit lower current penetration but show potential due to increasing ADHD awareness.
- Expanding into school-based health programs and adult ADHD clinics can elevate sales outside North America.
Conclusions
Focalin XR maintains a significant position in the ADHD treatment market with stable sales projections, despite patent expirations and competition from generics. Growth relies on expanding indications, market penetration, and maintaining prescriber preference amid competitive dynamics.
Key Takeaways
- Focalin XR's 2022 global sales are around $1.3 billion, with steady growth forecasted.
- North America, especially the U.S., accounts for over 65% of sales.
- Market share stands at approximately 8-10%; competitors like Adderall XR and Concerta dominate.
- Sales are expected to reach $180-200 million by 2025 and over $250 million by 2030.
- Risks include patent expiry effects, regulatory pressures, and market competition.
FAQs
1. How does Focalin XR compare in efficacy to its competitors?
Clinical studies show similar efficacy among stimulant ADHD medications; Focalin XR may have a slightly lower propensity for side effects like sleep disturbances, influencing prescriber preference.
2. What regulatory approvals are relevant for Focalin XR's expansion?
In the U.S., FDA approval covers ADHD in children and adults. Additional approvals for non-ADHD indications are limited but could influence market size if obtained.
3. How do generic versions impact Focalin XR's sales?
Generics, available since 2019, exert price competition but haven't significantly displaced the branded product due to trust and prescriber familiarity.
4. What are the typical reimbursement scenarios for Focalin XR?
Insurance coverage is generally favorable, with formulary inclusion common. Out-of-pocket costs vary based on insurance plans and co-pay policies.
5. What are the recent trends influencing ADHD medication prescriptions?
Increased diagnosis rates, especially among adults, and shifts toward non-stimulant options are notable trends potentially impacting Focalin XR's market position.