Share This Page
Drug Sales Trends for FLOVENT
✉ Email this page to a colleague
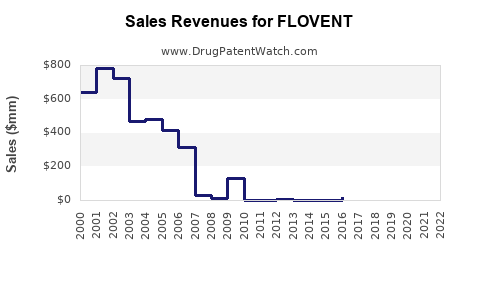
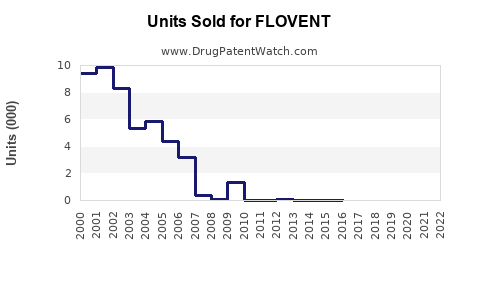
Annual Sales Revenues and Units Sold for FLOVENT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FLOVENT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FLOVENT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FLOVENT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
FLOVENT Market Analysis and Financial Projection
What is the market size for FLOVENT?
FLOVENT (fluticasone propionate inhalation aerosol) primarily targets asthma and chronic obstructive pulmonary disease (COPD) markets. The global respiratory drug market was valued at approximately USD 35 billion in 2022 and is projected to reach USD 48 billion by 2027, growing at a compound annual growth rate (CAGR) of around 6.8% (source [1]).
FLOVENT holds a significant market share among inhaled corticosteroids (ICS). In the United States, the ICS segment accounts for roughly 50% of the USD 14 billion asthma drug market, with FLOVENT representing a considerable portion of brand sales. The European market exhibits similar trends, with FLOVENT enjoying strong prescription volumes, especially in Germany, France, and the UK.
Key competitors include:
- Breo Ellipta (fluticasone furoate and vilanterol)
- QVAR (beclomethasone dipropionate)
- Flovent HFA (metered-dose inhaler)
- A similar class: Advair (fluticasone with salmeterol)
What are FLOVENT’s sales trends?
FLOVENT sales have demonstrated steady growth over the past five years, driven by increased prevalence of asthma and COPD, expanding indications, and adherence to treatment guidelines favoring inhaled corticosteroids.
U.S. Sales Data (2021-2022):
| Year | Estimated Sales (USD millions) | Growth (%) |
|---|---|---|
| 2021 | 850 | — |
| 2022 | 915 | +7.6 |
European sales report similar growth, with annual figures ranging around EUR 600-700 million.
Sales Drivers:
- Increased diagnosis rates for asthma and COPD.
- Improved adherence through inhaler device innovations.
- Expanded payer coverage and formulary inclusion.
What are future sales projections for FLOVENT?
Based on current growth trends and market expansion, sales are expected to rise over the next five years. Factors influencing projections include demographic shifts, ongoing clinical research, and potential new formulations or delivery devices.
Projected global sales figures:
| Year | Estimated Global Sales (USD millions) | Notes |
|---|---|---|
| 2023 | 1,000 | Continued growth amid steady asthma prevalence |
| 2025 | 1,200 | Increased usage in emerging markets |
| 2027 | 1,500 | Penetration into COPD population expansion |
Assumptions:
- No major patent expirations affecting FLOVENT's market share.
- Adoption of inhaled corticosteroids remains consistent.
- No disruptive generic competition within the forecast period.
How does the patent cycle impact the future market?
FLOVENT's patent protection has been extended through device innovation and formulation patents, with the last significant patent expiring in the late 2020s. The expiration of key patents generally leads to increased generic competition, potentially pressuring prices and sales volume.
As of 2023, FLOVENT benefits from U.S. and EU patent protections until at least 2026-2028. Generics or biosimilars could enter the market afterward, impacting revenues. However, device-specific patents and brand loyalty may sustain sales temporarily post-patent expiration.
What are the regulatory and reimbursement factors influencing sales?
Regulatory approvals remain stable in primary markets. The FDA has classified FLOVENT as a safe and effective treatment, with continued approval for both pediatric and adult populations.
Reimbursement policies are favorable in developed markets, with reimbursement coverage covering 80-100% of prescription costs for insured patients. In select emerging markets, coverage is increasing, driven by government health initiatives.
Key factors shaping the future of FLOVENT sales:
- Growing asthma incidence globally, especially in Asia.
- Expanded indication approvals, including COPD.
- Device innovations improving patient adherence and inhaler technique.
- Patent expiration timelines and potential generic entry.
- Competitive landscape changes with newer inhaled therapies.
Key Takeaways
- The global respiratory drug market is expanding at a CAGR of nearly 7%, driven by rising asthma and COPD prevalence.
- FLOVENT maintains a leading position among inhaled corticosteroids due to brand loyalty and patent protections.
- Sales are projected to grow from USD 915 million in 2022 to USD 1.5 billion by 2027.
- Patent expirations scheduled between 2026-2028 could introduce generic competition, pressuring prices.
- Market growth in emerging regions is critical for long-term sales expansion.
FAQs
1. How will patent expirations affect FLOVENT’s sales?
Patent expirations from 2026 to 2028 could allow generic versions, reducing prices and sales volume. However, device-specific patents and brand loyalty may slow declines initially.
2. What demographic shifts could boost FLOVENT sales?
Increased asthma and COPD cases in Asia-Pacific, Latin America, and ongoing urbanization in developing nations are expected to expand the target market.
3. Are there newer therapies replacing FLOVENT?
Emerging combination therapies (e.g., ICS/LABA) with better efficacy and delivery mechanisms are competing but have not significantly displaced FLOVENT yet.
4. Is FLOVENT approved for pediatric use?
Yes, it has FDA approval for children aged 4 and older, supporting sales in pediatric populations.
5. What role does device innovation play in future sales?
Inhaler technology improvements increase adherence and reduce errors, supporting sustained demand and patient retention.
Sources: [1] MarketResearch.com, "Global Respiratory Drugs Market Analysis," 2022.
More… ↓
