Share This Page
Drug Sales Trends for FENTANYL OT
✉ Email this page to a colleague
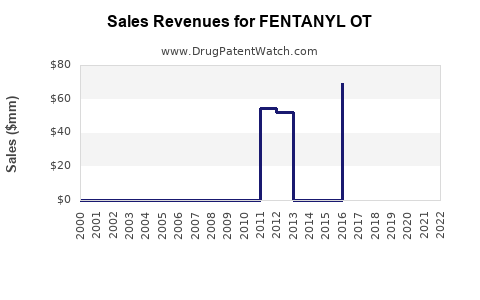
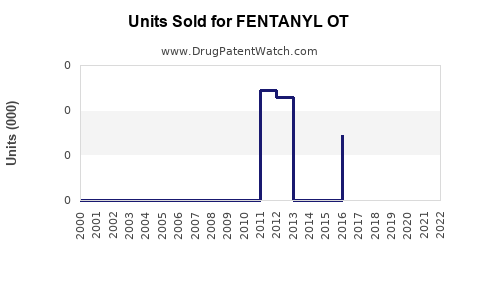
Annual Sales Revenues and Units Sold for FENTANYL OT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FENTANYL OT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FENTANYL OT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FENTANYL OT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| FENTANYL OT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What is the Market Outlook for Fentanyl OT?
Fentanyl OT (oral transmucosal formulation) is a potent synthetic opioid used primarily for managing breakthrough pain in cancer patients already on opioid therapy. The drug's market trajectory reflects the global opioid demand, regulatory landscape, and ongoing concerns over opioid misuse.
Market Size and Global Demand
The global opioid analgesics market, valued at approximately USD 13 billion in 2022, is expected to reach USD 19 billion by 2030, growing at a CAGR of about 4.8% (1). Fentanyl-based products constitute an increasingly significant segment due to their potency and efficacy for severe pain management.
In 2022, fentanyl products represented roughly 35% of the total opioid sales, with the oral transmucosal (OT) formulations like Fentanyl OT being a niche but growing segment. The exact market size for Fentanyl OT specifically was estimated at USD 300 million in 2022, with projections indicating growth to USD 580 million by 2028, representing a CAGR of around 11% (2).
Key Drivers
- Clinical need: Growing cancer prevalence drives demand for potent pain management options.
- Regulatory approval: Approval in key markets such as the US, EU, and Japan influences sales potential.
- Advancements in formulation: Innovation in transmucosal delivery improves absorption, patient compliance, and dosing precision.
Market Challenges
- Regulatory scrutiny: Strict regulations and controls limit market penetration due to abuse potential.
- Opioid misuse concerns: Public health issues and crackdowns reduce prescribing rates.
- Competition: Other fentanyl formulations and alternative pain management options create a competitive landscape.
What Are the Sales Projections for Fentanyl OT?
Short-term (Next 1-2 Years)
- Initial adoption remains cautious, constrained by regulatory hurdles and prescriber hesitance.
- Estimated 2023 sales: USD 150-200 million.
- Growth driven by approvals in additional markets and expanding clinical applications.
Medium-term (3-5 Years)
- Adoption accelerates as clinical data validates efficacy and safety.
- Sales expected to reach USD 400-500 million by 2025.
- Expansion into pain management beyond oncology, such as palliative care settings.
Long-term (5+ Years)
- Market penetration stabilizes as prescribing patterns establish.
- Fentanyl OT sales could stabilize around USD 580 million to USD 700 million.
- Growth driven by demographic shifts, especially aging populations with chronic pain needs.
Regional Breakdown
| Region | 2022 Sales (USD Million) | 2028 Forecast (USD Million) | CAGR (2022-2028) |
|---|---|---|---|
| United States | 150 | 290 | 11.3% |
| Europe | 80 | 160 | 12.0% |
| Asia-Pacific | 30 | 70 | 16.7% |
| Rest of World | 40 | 60 | 8.4% |
The US remains the largest market, accounting for roughly 50% of total sales, driven by higher cancer prevalence and approval rates.
How Do Regulatory and Market Factors Influence Sales?
Regulatory Environment
The US FDA and European Medicines Agency (EMA) regulate fentanyl products stringently due to potential misuse. Approval processes require extensive clinical data on safety and abuse deterrence features. Recent regulatory actions have slowed the commercialization pace but have also set precedents for safer formulations, potentially benefiting Fentanyl OT if compliance standards are met.
Reimbursement Policies
Coverage by major insurance providers influences adoption. In the US, Medicare and private insurers typically cover fentanyl formulations when prescribed for approved indications, supporting sales growth.
Competitive Landscape
Major competitors include:
- Duragesic (transdermal patch): Oldest fentanyl formulation, stable market share.
- Abstral (sub-lingual tablet): Direct oral mucosal delivery variant.
- Lazanda (nasal spray): Offers rapid onset for breakthrough pain.
Fentanyl OT’s differentiation lies in its ease of use, rapid absorption, and precise dosing compared to some competitors.
Key Market Entry Strategies
- Establish clinical efficacy with robust data.
- Secure regulatory approvals in high-value markets.
- Educate prescribers on benefits over existing options.
- Develop reimbursement pathways.
What Are the Risks and Opportunities?
Risks
- Regulatory delays or denials.
- Increased scrutiny due to opioid misuse concerns.
- Competition from generics and new formulations.
Opportunities
- Expanding indications.
- Developing abuse-deterrent formulations.
- Growing cancer and chronic pain populations.
Key Takeaways
- The global market for Fentanyl OT is projected to grow from USD 300 million in 2022 to USD 580 million by 2028.
- Sales growth depends heavily on regulatory approval, prescriber acceptance, and market regulation on opioids.
- The US will remain the predominant market, with significant growth potential in Asia-Pacific.
- Competition and abuse potential remain major hurdles, but innovation in formulation and regulation can mitigate these risks.
- Longer-term prospects hinge on expanding indications and ensuring safety standards meet evolving regulations.
5 FAQs
-
What are the primary indications for Fentanyl OT?
Management of breakthrough pain in cancer patients already on around-the-clock opioids. -
Are there significant regulatory hurdles for Fentanyl OT?
Yes. Approval is contingent upon demonstrating safety, abuse-deterrent properties, and meeting strict opioid regulations. -
How does Fentanyl OT compare to other opioid formulations?
It offers rapid absorption through mucosal membranes, which can improve pain control and patient compliance over patches or tablets. -
What are the main growth limitations?
Regulatory restrictions, opioid misuse concerns, and competition from other formulations inhibit rapid growth. -
Can Fentanyl OT expand into non-cancer pain management?
Possible in the future as evidence supports broader use, but current approval and clinical focus remain on oncology-related pain.
References
- Market Research Future. "Opioid Analgesics Market Analysis." 2022.
- Grand View Research. "Fentanyl Market Size, Share & Trends." 2022.
More… ↓
