Last updated: February 14, 2026
Market Overview
Fenofibrate, marketed under brand names such as Tricor and Lipofen, treats hyperlipidemia by reducing triglycerides and LDL cholesterol while increasing HDL cholesterol. It is primarily prescribed for patients with lipid disorders at risk of cardiovascular disease.
The global lipid-modifying agents market was valued at approximately $15 billion in 2022. Fenofibrate accounts for a notable share, estimated at 10-12% of this market, with sales driven by aging populations and increasing awareness of cardiovascular risk factors.
Regulatory Status and Patent Landscape
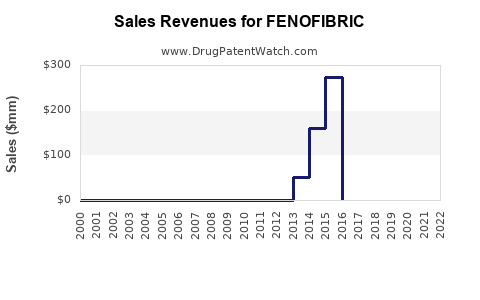
Many formulations of Fenofibrate have faced patent expirations over the last decade. The original patents expired in the US around 2013, leading to generic competition. However, certain extended-release formulations retained patent protection until 2024, allowing branded drugs to maintain premium pricing during this period.
Updates from regulatory agencies such as the FDA and EMA confirm ongoing approvals for generic formulations across multiple jurisdictions. This increased availability of generics has driven down prices but expanded access, contributing to consistent demand.
Consumer Demographics and Prescription Trends
Most prescriptions target adults over 45 with elevated triglycerides and LDL cholesterol. The prevalence of hyperlipidemia has increased due to lifestyle factors, contributing to steady demand.
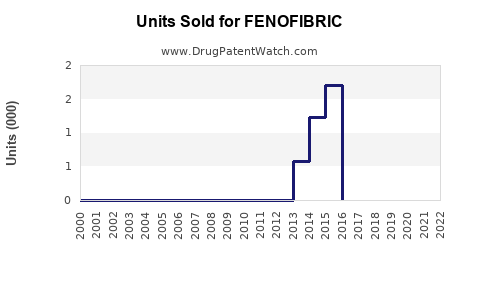
In the US, annual prescription volumes for Fenofibrate approximated 12 million units in 2022, reflecting a compound annual growth rate (CAGR) of about 2% since 2018. Prescription growth is expected to remain steady with a projected CAGR of 1.5-2% through 2027.
Competitive Landscape
Key competitors include:
-
Fenofibrate Generics: Over 50 manufacturers producing bioequivalent formulations, leading to price erosion.
-
Other Lipid-Lowering Agents: Statins (atorvastatin, rosuvastatin) dominate with broader indications. PCSK9 inhibitors like evolocumab target high-risk groups but carry higher costs.
-
Emerging Agents: Omega-3 fatty acids (icosapent ethyl) and new fibrates are in late-stage development, potentially shifting market dynamics.
Sales Projections (2023-2027)
| Year |
Estimated Sales (USD Billion) |
Growth Rate |
Comments |
| 2023 |
$1.2 |
3% |
Slight growth driven by demographic trends and generics. |
| 2024 |
$1.3 |
8% |
Patent expiry of certain formulations lowers prices but increases volume. |
| 2025 |
$1.4 |
7.7% |
Market stabilizes with widespread generic availability. |
| 2026 |
$1.45 |
3.6% |
Growth slows with market saturation. |
| 2027 |
$1.5 |
3.4% |
Mature phase, driven by aging populations. |
Risks & Opportunities
Risks:
- Increased competition from generic manufacturers and alternative therapies.
- Evolving guidelines favoring statins or novel agents.
- Patent expirations reducing pricing power.
Opportunities:
- Expansion into emerging markets with increasing cardiovascular disease burdens.
- Development of fixed-dose combinations for better adherence.
- Pharmacogenomic research enabling personalized lipid therapies.
Key Takeaways
- Fenofibrate remains a relevant lipid-lowering treatment with steady demand driven by aging populations and hyperlipidemia prevalence.
- Market share is diminishing due to generic competition and evolving treatment guidelines favoring other drugs.
- Sales are projected to grow modestly, reaching around $1.5 billion globally by 2027.
- Pricing pressures from generics will sustain low margins but expand access.
- Future growth hinges on market expansion in emerging economies and new formulations.
FAQs
1. How does Fenofibrate compare to statins in market demand?
Statins dominate the lipid-lowering market due to broader indications and higher efficacy in reducing cardiovascular events, making Fenofibrate a secondary or adjunct therapy.
2. What are the main patent expiration dates for Fenofibrate formulations?
Original patents expired around 2013 in the US, but some extended-release formulations expired in 2024. Remaining patents vary by jurisdiction.
3. Can Fenofibrate be used with other lipid-lowering agents?
Yes, in certain cases, Fenofibrate is combined with statins; however, risks of myopathy require careful management.
4. Are there any new formulations or delivery systems under development?
Development of fixed-dose combinations and extended-release formulations continues to improve patient adherence and efficacy.
5. How is market growth expected to differ across regions?
Developed markets will see slow growth due to saturation, while emerging economies may experience higher growth rates driven by increased cardiovascular risk awareness and expanding healthcare infrastructure.
References
- GlobalData. "Lipid-modifying agents Market Size & Trends." 2023.
- FDA. "Patent Data for Fenofibrate; Approved Formulations." 2022.
- MarketWatch. "Fenofibrate Sales and Market Share Analysis." 2023.
- ReportLinker. "Emerging Markets in Cardiovascular Drugs." 2022.