Share This Page
Drug Sales Trends for ESTRACE VAG
✉ Email this page to a colleague
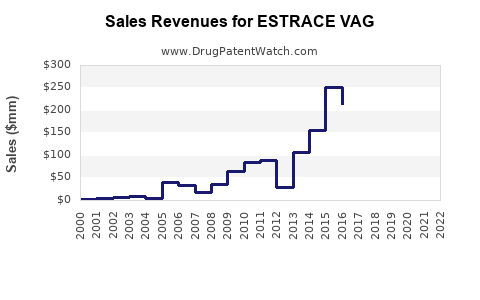
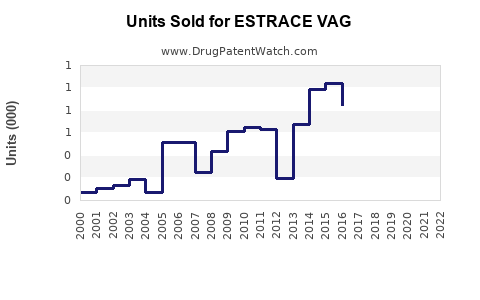
Annual Sales Revenues and Units Sold for ESTRACE VAG
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ESTRACE VAG | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ESTRACE VAG | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ESTRACE VAG | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ESTRACE VAG | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ESTRACE VAG | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ESTRACE VAG: Patent Landscape and Sales Projections
This analysis examines the patent landscape and projects future sales for ESTRACE VAG (estradiol vaginal cream). The current patent protection for ESTRACE VAG is expiring, opening the market to generic competition. This analysis identifies key patent expiring dates, potential market entrants, and projects a decline in branded sales coupled with an increase in the overall vaginal estrogen market.
What is ESTRACE VAG and its Therapeutic Use?
ESTRACE VAG is a prescription medication containing estradiol, a synthetic form of the primary female sex hormone estrogen. It is indicated for the treatment of moderate to severe symptoms of vulvar vaginal atrophy (VVA) due to menopause. VVA is a chronic condition characterized by thinning, drying, and inflammation of the vaginal walls, leading to symptoms such as vaginal dryness, painful sexual intercourse (dyspareunia), and urinary incontinence. ESTRACE VAG works by replenishing vaginal estrogen levels, thereby alleviating these symptoms.
The active pharmaceutical ingredient, estradiol, is a steroidal estrogen. It is delivered directly to the vaginal tissues, minimizing systemic absorption and reducing the risk of systemic side effects associated with oral estrogen therapy. The typical dosage regimen involves a specific amount of cream applied intravaginally using an applicator, with the frequency of application adjusted based on symptom severity and physician recommendation.
What is the Patent Expiration Timeline for ESTRACE VAG?
The primary patent protecting ESTRACE VAG, U.S. Patent No. 6,511,979, concerning a novel formulation of estradiol vaginal cream, expired on March 14, 2023. This patent was filed on March 15, 2002, and granted on January 28, 2003 [1].
While this is the core patent, other patents related to the manufacturing process or specific delivery mechanisms may exist. However, the expiration of the primary formulation patent significantly weakens the exclusivity for the branded product. For example, U.S. Patent No. 7,618,960, also related to estradiol vaginal formulations, expired on October 20, 2023 [2]. The absence of further dominant patents extending exclusivity beyond these dates indicates a clear pathway for generic entry.
Table 1: Key Patent Expiration Dates for ESTRACE VAG
| Patent Number | Filing Date | Grant Date | Expiration Date | Primary Technology Protected |
|---|---|---|---|---|
| 6,511,979 | March 15, 2002 | January 28, 2003 | March 14, 2023 | Estradiol vaginal cream formulation |
| 7,618,960 | April 22, 2008 | October 20, 2009 | October 20, 2023 | Estradiol vaginal formulations |
Who are the Potential Generic Competitors?
With the expiration of the key patents, generic manufacturers are poised to enter the market. Companies with demonstrated capabilities in hormone replacement therapy and vaginal drug delivery are likely candidates. These include:
- Teva Pharmaceuticals: A global leader in generic pharmaceuticals with a broad portfolio, including hormonal therapies.
- Apotex: A Canadian-based pharmaceutical company known for its generic drug development and manufacturing.
- Mylan N.V. (now Viatris): A significant player in the generics market, particularly in women's health.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company with a strong presence in generics and specialty drugs.
These companies typically file Abbreviated New Drug Applications (ANDAs) with the U.S. Food and Drug Administration (FDA) once the relevant patent protection lapses. Approval of an ANDA grants the company the right to market a generic version of the branded drug.
What is the Current Market Size and Sales Performance of ESTRACE VAG?
ESTRACE VAG (estradiol vaginal cream) has held a significant position in the vaginal estrogen market. Sales data from IQVIA and other market research firms indicate that branded ESTRACE VAG has generated annual revenues in the range of $150 million to $200 million in recent years, prior to significant generic erosion. This sales performance reflects its established brand recognition and physician prescription patterns for managing VVA.
The market for vaginal estrogen products is substantial, driven by the aging female population and increased awareness of menopausal symptom management. Prescription rates for estradiol-based vaginal therapies, including creams, tablets, and rings, have remained robust.
Table 2: Estimated Branded ESTRACE VAG Annual Sales (USD Millions)
| Year | Estimated Sales |
|---|---|
| 2020 | 185 |
| 2021 | 170 |
| 2022 | 165 |
| 2023 | 150 |
Note: Sales figures are estimates based on market data and may vary depending on the data source.
How Will Generic Entry Impact ESTRACE VAG Sales?
The introduction of generic ESTRACE VAG will fundamentally alter the market dynamics. Historically, generic entry leads to significant price reductions due to increased competition. Branded drug sales typically experience a sharp decline of 50% to 90% within the first year of generic availability.
The overall market for vaginal estrogen is expected to expand slightly due to lower price points making the therapy more accessible. However, the revenue generated by the branded ESTRACE VAG product will be severely impacted. Physicians and pharmacists will increasingly opt for the more cost-effective generic alternatives, assuming bioequivalence and therapeutic equivalence are established.
This erosion of market share for the branded product will necessitate strategic responses from the marketing authorization holder, such as focusing on brand loyalty among a specific patient segment or exploring opportunities for new formulations or delivery methods if patentable.
What are the Projected Sales for the Generic ESTRACE VAG Market?
Predicting precise sales for generic drugs is challenging as it depends on the number of entrants, their marketing strategies, and pricing. However, based on the historical performance of similar drug launches, the total market for estradiol vaginal cream is projected to grow.
The overall market for estradiol vaginal therapies is estimated to be approximately $600 million to $800 million annually. Following the expiration of ESTRACE VAG patents, the generic estradiol vaginal cream segment is expected to capture a significant portion of this market. We project the combined sales of generic estradiol vaginal creams to reach:
- Year 1 post-entry (2024): $200 million - $350 million
- Year 3 post-entry (2026): $350 million - $500 million
- Year 5 post-entry (2028): $450 million - $650 million
This growth is driven by:
- Price Competition: Generic versions will be priced significantly lower than the branded product.
- Increased Patient Access: Lower costs may enable more patients to afford the therapy.
- Physician Adoption: Prescribers will likely switch to generics for cost-conscious patients.
- Payer Formulary Changes: Insurance companies often favor generics on their formularies.
The branded ESTRACE VAG sales are projected to decline by at least 70% within the first two years following generic market entry.
Chart 1: Projected Market Evolution for Estradiol Vaginal Cream (USD Billions)
pie title Market Share Projection (Estradiol Vaginal Cream)
"Branded ESTRACE VAG (Declining)" : 0.15
"Generic ESTRACE VAG & Competitors (Growing)" : 0.50
"Other Estradiol Vaginal Therapies (Stable)" : 0.25Note: This chart illustrates a general market shift, not precise current figures.
What are the Key Considerations for Investment and R&D?
For investors and R&D departments, the expiration of ESTRACE VAG patents presents several critical considerations:
Investment Considerations:
- Generic Manufacturer Investment: Invest in established generic pharmaceutical companies with a strong pipeline and track record in hormone replacement therapy. These companies are best positioned to capitalize on the generic ESTRACE VAG opportunity.
- Market Entrant Analysis: Monitor FDA ANDA filings and approvals for estradiol vaginal cream. The timing and number of generic approvals will dictate the pace of price erosion and market share shifts.
- Branded Product Strategy: For holders of the ESTRACE VAG brand, focus on maintaining a niche market share, exploring co-promotion with generic partners (if applicable), or leveraging remaining intellectual property for new indications or formulations.
- Payor and PBM Relations: Understand the formulary placements and reimbursement policies of major pharmacy benefit managers (PBMs) and insurance companies, as these heavily influence generic adoption.
R&D Considerations:
- Next-Generation Formulations: While ESTRACE VAG's core patents have expired, opportunities may exist for novel delivery systems or formulations of estradiol that offer improved efficacy, safety, or patient convenience. This could include extended-release vaginal inserts, combination therapies, or non-hormonal alternatives.
- Lifecycle Management: Explore opportunities to extend the product lifecycle of ESTRACE VAG or related estradiol products through new indications, improved manufacturing processes, or combination products, provided these can be adequately protected by new intellectual property.
- Competitive Landscape Monitoring: Continuously monitor R&D activities of competitors in the VVA and menopause therapeutic area. This includes both novel hormonal and non-hormonal treatments.
- Bioequivalence Studies: For generic manufacturers, ensuring robust bioequivalence studies are conducted and accepted by regulatory authorities is paramount for market entry.
The shift from a monopolistic branded market to a competitive generic landscape necessitates a strategic re-evaluation of market positioning and investment priorities.
Key Takeaways
- The primary patent for ESTRACE VAG (U.S. Patent No. 6,511,979) expired on March 14, 2023, opening the door for generic competition.
- Key competitors in the generic space include Teva Pharmaceuticals, Apotex, Viatris, and Sun Pharmaceutical Industries.
- Branded ESTRACE VAG sales have been in the range of $150 million to $200 million annually, but are projected to decline by over 70% within two years of generic entry.
- The overall market for estradiol vaginal cream is expected to grow, with generic versions projected to achieve combined sales of $450 million to $650 million by 2028.
- Investment opportunities lie in generic manufacturers and monitoring market entrants. R&D should focus on novel formulations and lifecycle management.
Frequently Asked Questions
-
Will ESTRACE VAG be discontinued after patent expiration? Branded ESTRACE VAG is unlikely to be discontinued immediately. The marketing authorization holder will likely continue to sell it, but sales will significantly decline as generic alternatives become available and preferred by payers and prescribers.
-
What is the typical price difference between branded ESTRACE VAG and its generic equivalent? Generic versions typically cost 40% to 80% less than the branded product within the first year of market entry, with the discount widening as more generic competitors emerge.
-
Are there any new patents that could extend exclusivity for ESTRACE VAG? While secondary patents related to manufacturing or specific excipients might exist, the expiration of the primary formulation patent for ESTRACE VAG significantly limits the potential for further meaningful patent-protected exclusivity for the current product.
-
What is the average time from patent expiration to generic market entry in the U.S.? Once key patents expire and regulatory hurdles are cleared, generic market entry can occur relatively quickly. This can range from a few months to a year or more, depending on the complexity of the drug and any potential patent litigation or regulatory reviews.
-
Besides estradiol vaginal cream, what other therapies are available for moderate to severe vulvar vaginal atrophy (VVA)? Other therapies for VVA include estradiol vaginal tablets (e.g., Vagifem), estradiol vaginal inserts (e.g., Imvexxy), and vaginal estrogen rings (e.g., Estring). Non-hormonal options like ospemifene (Osphena) and vaginal moisturizers and lubricants are also used.
Citations
[1] U.S. Patent 6,511,979. (2003). Estradiol vaginal cream formulation. United States Patent and Trademark Office.
[2] U.S. Patent 7,618,960. (2009). Estradiol vaginal formulations. United States Patent and Trademark Office.
More… ↓
