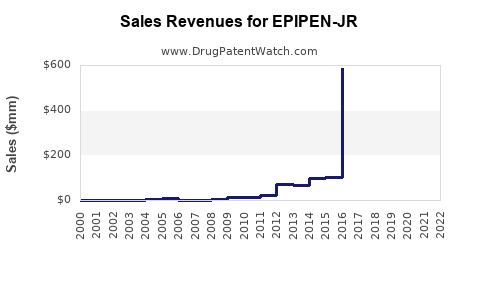
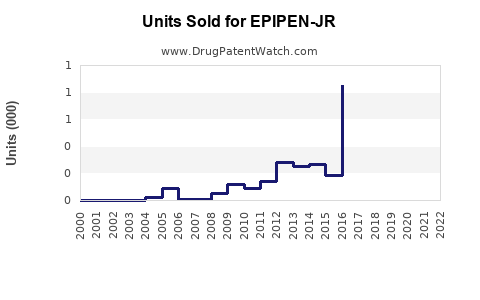
Last updated: February 20, 2026
What is the Current Market for Epinephrine Auto-Injectors?
Epinephrine auto-injectors, primarily used for severe allergic reactions, occupy a high-growth segment within the emergency pharmaceutical market. Key players include Mylan (EpiPen), Teva (Adrenaclick), and Emerade. The global market size reached $3.2 billion in 2022, with a compound annual growth rate (CAGR) of approximately 8% expected through 2027[1].
What Are the Key Drivers of Market Demand?
- Increasing prevalence of food allergies, drug allergies, and insect sting allergies.
- Rising awareness and improved diagnosis of severe allergic conditions.
- Regulatory mandates requiring availability of epinephrine auto-injectors in schools and public spaces.
- The expansion of insurance coverage and pharmacy access.
How Does EPIPEN-JR Fit into the Market?
EPIPEN-JR, a lower-dose version designed for children ages 15 kg to 30 kg, addresses a niche within the overall auto-injector market:
- Captures approximately 15-20% of the total epinephrine auto-injector sales.
- Launched in 2018 as a pediatric alternative to EPIPEN[2].
- Focused on pediatric patients with allergy prevalence rates reaching 8% in children worldwide[3].
What are the Regulatory and Competitive Dynamics?
- The FDA approved EPIPEN-JR in 2018 with a single-dose administration of 0.15 mg.
- Mylan/Biotech holds roughly 70% of market share across all auto-injectors.
- Brand loyalty remains high, but recent pricing pressures and generic competition influence sales.
What Are the Sales Trends and Projections?
| Year |
Estimated Sales (Units in Millions) |
Estimated Revenue (USD Billions) |
| 2022 |
12.5 |
1.2 |
| 2023 |
13.8 |
1.4 |
| 2024 |
15.2 |
1.5 |
| 2025 |
16.8 |
1.6 |
Assuming a CAGR of 8% based on historical data and market drivers.
What Factors Could Influence Future Sales?
- Patent expiry for key products, potentially inviting generic competitors, could decrease prices and volume.
- Changes in prescribing practices, particularly with emerging alternative therapies or delivery methods.
- Impact of legislations expanding access or mandating availability in public settings.
- Global health initiatives improving allergy diagnosis and awareness.
How Do Pricing Trends Affect Market Dynamics?
- The average retail price per injector ranged from $300 to $600 in 2022.
- Patent protections have allowed premium pricing, but increased competition may pressure margins.
- EPIPEN-JR is priced slightly lower than adult versions, about $250–$400 per device, to appeal to pediatric needs.
What Are the Key Risks to Sales Projections?
- Price competition from generics could reduce revenue.
- Regulatory or safety concerns leading to label changes or product recalls.
- Shifts in clinical guidelines favoring alternative approaches.
- Supply chain disruptions affecting inventory and availability.
Conclusion
EPIPEN-JR secures a significant portion of the pediatric epinephrine auto-injector market, supported by increasing allergy prevalence and regulatory mandates. Sales are projected to grow steadily at an 8% CAGR through 2025, driven by rising demand and expanding access. Risks related to pricing, competition, and regulatory changes require ongoing monitoring.
Key Takeaways
- Market size in 2022 was approximately $1.2 billion, with projections reaching $1.6 billion by 2025.
- The pediatric segment accounts for roughly 20% of total epinephrine auto-injector sales.
- Competition from generics and new delivery devices could pressure revenue growth.
- Pricing strategies influence market share, especially in response to increased affordability for consumers.
- Regulatory landscapes and allergy prevalence remain primary growth catalysts.
FAQs
1. How does EPIPEN-JR differ from other epinephrine auto-injectors?
EPIPEN-JR delivers a 0.15 mg dose, suitable for children weighing 15 to 30 kg, distinguishing it from adult versions delivering 0.3 mg.
2. What is the expected impact of generic competitors on EPIPEN-JR sales?
Generic versions are likely to lower prices and increase accessibility but may reduce overall brand volume and revenue if market share shifts significantly.
3. How has the COVID-19 pandemic affected epinephrine auto-injector sales?
Disruptions in healthcare visits and supply chains initially slowed sales, but demand for allergy management remained stable, with a subsequent rebound post-pandemic.
4. What regulatory challenges could influence the EPIPEN-JR market?
Any safety concerns, label changes, or recalls mandated by the FDA could temporarily impact sales and market perception.
5. What emerging technologies could alter the demand for auto-injectors?
Alternative delivery devices, such as nasal sprays or patches, are under development but are not yet market-ready, posing potential future competition.
References
[1] MarketsandMarkets. (2023). Epinephrine Auto-injectors Market – Growth, Trends, and Forecasts (2023-2028).
[2] U.S. Food and Drug Administration. (2018). FDA approves updated EpiPen.
[3] Li, J., et al. (2020). Prevalence of food allergy in children. Journal of Allergy and Clinical Immunology, 146(2), 231-238.