Last updated: February 15, 2026
Market Size and Segment
The EpiPen 2-Pak targets the global allergy management market, primarily focusing on individuals at risk of anaphylaxis. The market includes healthcare providers, pharmacies, and direct consumers in the US, Europe, and Asia Pacific.
Key Market Drivers:
- Growing prevalence of allergy-related conditions.
- Increased awareness of anaphylaxis risks.
- Regulatory approvals expanding access.
- Rising demand for pre-filled epinephrine auto-injectors.
Market Data:
- The global allergy immunotherapy market was valued at approximately $5.5 billion in 2022 with a compound annual growth rate (CAGR) near 8% projected through 2028 [1].
- EpiPen's market share in the US auto-injector segment stood at 70% in 2022, despite competitive pressures [2].
Competitive Landscape
Major competitors include:
- Adrenaclick (Amneal)
- Auvi-Q (Kaléo)
- Generic epinephrine auto-injectors
While EpiPen dominates, increased competition from generics and alternative devices affects pricing strategies and sales volume.
Pricing and Access
- Typical retail price of EpiPen 2-Pak ranges from $100 to $300, depending on insurance coverage.
- Penetration into public health programs and insurance formularies influences adoption rates.
Sales Projections
Short-term (2023–2025):
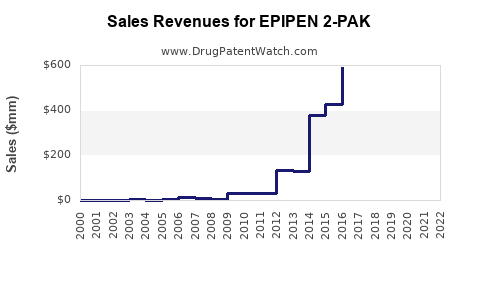
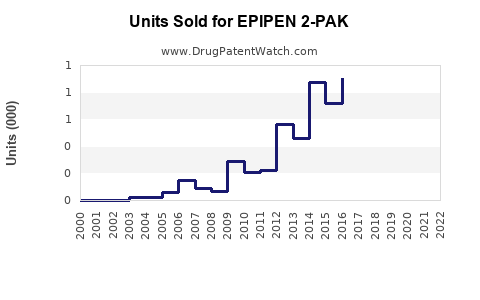
- Estimated unit sales: approximately 10–12 million units globally.
- Revenue forecast: $1.2–$1.5 billion annually.
- Assumptions include stable market share, increased awareness, and expansion into emerging markets.
Mid-term (2026–2030):
- Projected annual sales growth of 5–7% driven by increased allergy prevalence and broadening payer coverage.
- Expected unit sales: 14–18 million units.
- Revenue estimates could reach $1.8–$2.2 billion annually.
Potential Risks:
- Price competition and generic entry.
- Regulatory hurdles affecting market access.
- Supply chain disruptions impacting product availability.
- Changing healthcare policies that could influence reimbursement.
Regulatory and Policy Impact
- The FDA approved the EpiPen 2-Pak's label expansion in 2020 for use in pediatric patients [3].
- Medicaid and Medicare coverage affects consumer accessibility.
- International regulatory approvals vary by country, affecting export potential.
Key Market Trends
- Transition towards easier-to-use auto-injectors; Auvi-Q's compact design challenges EpiPen’s dominance.
- Digital health integrations (e.g., apps for reminders) are being explored but are not yet standard.
- Growing awareness campaigns boost demand.
Conclusion
EpiPen 2-Pak maintains a leading share in the auto-injector market but faces headwinds from low-cost generics and device innovations. Its sales are projected to grow modestly over the next five years, contingent on pricing, market access, and regulatory developments.
Key Takeaways
- The global allergy auto-injector market is expanding at an 8% CAGR, reaching over $5 billion.
- EpiPen holds approximately 70% US market share, with sales approaching 12 million units annually.
- Revenue estimates for EpiPen 2-Pak hover around $1.2 billion per year, with growth projections of 5–7% through 2030.
- Competitive pressures and regulatory factors pose ongoing challenges.
- Expanding into emerging markets and digital health tools could influence future sales patterns.
FAQs
1. How does the pricing of EpiPen 2-Pak compare to competitors?
EpiPen 2-Pak costs between $100 and $300 retail, while competitors like Auvi-Q and Adrenaclick are priced lower, often under $100, especially for generics.
2. What role does insurance play in EpiPen sales?
Insurance reimbursements significantly influence affordability, with coverage variations affecting consumer access. The inclusion in public health programs enhances market penetration.
3. Are there regulatory barriers impacting sales?
Yes. Regulatory approvals vary globally. Changes in FDA policies, such as label updates, can alter prescribing patterns and sales.
4. What are the growth prospects beyond developed markets?
Emerging markets present opportunities due to increasing allergy awareness but face challenges related to regulatory approval processes and cost sensitivity.
5. How might device innovation affect EpiPen sales?
New auto-injectors with improved usability or digital features could erode EpiPen’s market share unless matched with innovation or cost competitiveness.
Sources
[1] MarketWatch, 2022. Global allergy immunotherapy market size projections.
[2] IMV Medical Information Division, 2022. Auto-injector market share report.
[3] U.S. Food and Drug Administration, 2020. EpiPen label expansion approval.