Last updated: February 20, 2026
Enbrel (etanercept) is a biologic TNF inhibitor primarily used to treat autoimmune conditions such as rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis. Market dynamics, competitive landscape, regulatory environment, and historical sales data shape its future sales expectations.
Market Overview
- Therapeutic Area: Immunology, with a focus on autoimmune inflammatory diseases.
- Indications: Rheumatoid arthritis (RA), psoriatic arthritis (PsA), ankylosing spondylitis (AS), plaque psoriasis (PsO), and juvenile idiopathic arthritis.
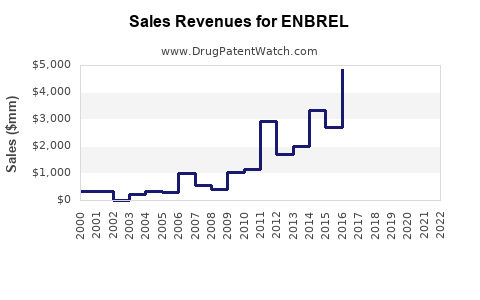
- Sales in 2022: Approx. $6.7 billion globally (EvaluatePharma, 2022).
- Market Share: Among TNF inhibitors, Enbrel holds roughly 25-30% of the global biologic TNF inhibitor market (IQVIA, 2022).
Competitive Landscape
| Drug |
Company |
Route of Administration |
Year Approved |
Global Sales 2022 |
Key Differentiator |
| Enbrel |
Amgen |
Subcutaneous |
1998 |
$6.7 billion |
First TNF inhibitor, broad approval |
| Humira |
AbbVie |
Subcutaneous |
2002 |
$21.1 billion |
Market leader, wider label expansion |
| Remicade |
Janssen/Johnson & Johnson |
IV infusion |
1998 |
Approx. $8 billion |
First infliximab, IV route |
| Cimzia |
UCB |
Subcutaneous |
2009 |
Approx. $2.2 billion |
Less frequent dosing |
Key Market Drivers
- Growing prevalence of autoimmune diseases.
- Expanding aging population.
- Increasing physician familiarity with biologics.
- Patent expiration for key competitors (Humira in 2023 in the US).
Patent and Regulatory Status
- Patent expiry for Enbrel was in 2028 in the US, with potential extensions or biosimilar entries possible.
- Amgen has engaged in biosimilar development, with biosimilars potentially impacting revenue starting mid-2028.
Sales Projection Assumptions
- Current sales: approximately $6.7 billion worldwide (2022).
- Average annual growth rate (2023-2027): 3-4% due to increasing autoimmune disease diagnoses but offset by biosimilar competition and market saturation.
- Post-2028 impact: biosimilar entry may reduce revenues by 40-60%, depending on market penetration and pricing strategies.
2023-2027 Sales Forecast
| Year |
Projected Sales (USD billion) |
Notes |
| 2023 |
$6.8 – $7.0 |
Slight growth driven by new indications and patient accumulation. |
| 2024 |
$7.0 – $7.2 |
Continued growth, stable market share. |
| 2025 |
$7.2 – $7.4 |
Growth slightly slows due to biosimilar competition. |
| 2026 |
$7.3 – $7.5 |
Market stabilizes; biosimilar impact increases. |
| 2027 |
$7.4 – $7.6 |
Biosimilar market entry expected, market share may decline. |
Post-Patent Biosimilar Entry Impact
| Year |
Estimated Revenue Impact |
Market Share Reduction |
Notes |
| 2028 |
Up to 50% reduction |
High |
Biosimilar uptake varies by region, significantly reduces sales. |
| 2029+ |
Stabilization at new market levels |
30-40% decline |
Continued biosimilar competition, possible patent litigations. |
Regional Outlook
| Region |
2022 Sales (USD billion) |
Growth Rate 2023-2027 |
Key Factors |
| North America |
$4.0 |
2-3% |
Largest market, high biologic adoption |
| Europe |
$2.2 |
2-3% |
Competitive biosimilar activity |
| Asia-Pacific |
$0.5 |
4-5% |
Growing access, expanding healthcare infrastructure |
Risks and Market Challenges
- Patent expiry and biosimilar entry could decrease sales.
- Price competition from biosimilars.
- Regulatory hurdles in emerging markets.
- Market saturation in developed regions.
- Patient switching behaviors influenced by biosimilar availability and healthcare policies.
Strategic Outlook
- Enbrel's revenue will likely peak around 2027 before biosimilar influence intensifies.
- Amgen may pursue lifecycle management strategies such as formulation improvements or label expansions.
- Market share may shift from originator to biosimilars, especially outside the U.S.
Key Takeaways
- Enbrel remains a leading biologic TNF inhibitor, with approximately $6.7 billion in sales in 2022.
- Growth is steady but limited by patent expiration and biosimilar competition.
- Post-2028 revenues are projected to decline notably with biosimilar market entry.
- Regional differences will influence the pace and extent of revenue declines.
- Strategic actions include diversifying indications and pursuing biosimilar market entry.
FAQs
1. When is Enbrel's patent expiration?
The patent in the U.S. expired in 2028, with potential extensions or legal challenges possible.
2. How will biosimilars affect Enbrel’s sales?
Biosimilars introduced post-2028 could reduce Enbrel’s sales by up to 50% within a few years, depending on market uptake.
3. What are Enbrel’s primary indications?
Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, plaque psoriasis, and juvenile idiopathic arthritis.
4. What competitive advantages does Enbrel hold?
First-mover advantage, broad indication approval, established physician familiarity.
5. Which regions are critical for Enbrel's future sales?
North America and Europe will see sales decline due to biosimilar entry, while Asia-Pacific offers growth opportunities.
References
[1] EvaluatePharma. (2022). World Preview 2022.
[2] IQVIA. (2022). The Global Use of Medicines Report.