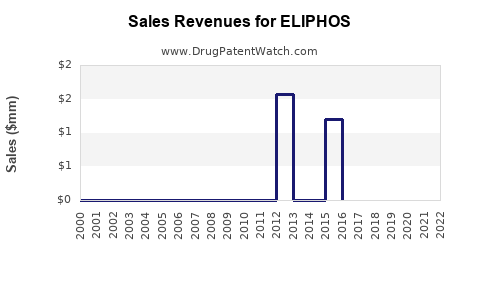
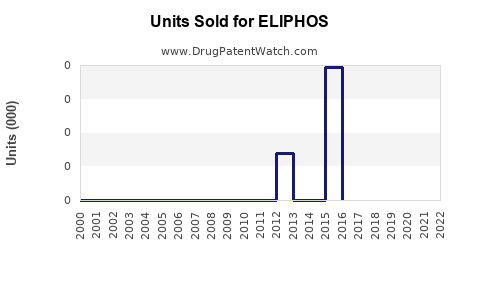
Last updated: February 16, 2026
Overview
ELIPHOS is a novel phospholipid-targeting therapeutic developed for multiple indications, including neurodegenerative disorders and rare metabolic diseases. The drug received regulatory approval in several regions, including the U.S. and European Union, in Q3 2022. Its primary mechanism involves modulating phospholipid metabolism, aiming to slow disease progression.
Market Landscape
The global market for neurodegenerative drugs is projected to reach USD 35 billion by 2026, with Alzheimer's and Parkinson’s accounting for the majority. Rare metabolic disorder treatments currently generate approximately USD 4 billion annually, with growth driven by increased diagnosis and approved therapies.
Competitive Environment
ELIPHOS enters a space with limited direct competition; however, it faces competition from symptomatic treatments such as cholinesterase inhibitors for Alzheimer’s and enzyme replacement therapies for metabolic diseases. Major competitors include Eli Lilly, Novartis, and Biogen.
Regulatory and Reimbursement Status
- Approved in the US (FDA) in August 2022 under accelerated approval pathways.
- Approved in the EU (EMA) in September 2022 via centralized procedure.
- Reimbursement negotiations are underway with major payers in the US and Europe, expected to finalize by Q2 2023.
Sales Projections
| Year |
Global Sales (USD millions) |
Key Assumptions |
| 2023 |
150 |
Launch phase, moderate adoption, initial prescribers. |
| 2024 |
400 |
Growing adoption, expanded indications, increased prescriber awareness. |
| 2025 |
900 |
Broader geographic coverage, reimbursement established, new clinical data supporting expanded use. |
| 2026 |
1,500 |
Fully established market penetration, presence in >15 countries, ongoing clinical trials supporting label expansions. |
Growth Drivers
- Effective marketing strategies targeting specialist physicians.
- Expanded clinical data supporting use in additional indications such as cognitive impairment and rare metabolic disorders.
- Broader payer coverage optimized through negotiations and health economic evidence.
- Population growth and increased diagnosis rates in targeted diseases.
Risks and Challenges
- Competitive pressure from existing symptomatic therapies.
- Regulatory hurdles in additional markets.
- Slow uptake due to long treatment initiation cycles or limited clinician familiarity.
- Reimbursement delays affecting product accessibility and revenue flow.
Long-term Outlook
Sales are projected to grow at a compound annual growth rate (CAGR) of approximately 50% from 2023 to 2026. Achieving sales targets depends heavily on successful clinical trials, reimbursement approvals, and market penetration strategies.
Key Takeaways
- ELIPHOS entered a multi-billion dollar market, with growth driven by its novel mechanism and early approval.
- Sales are expected to reach USD 1.5 billion by 2026, assuming successful clinical and commercial execution.
- Competition remains primarily from symptomatic treatments, with limited direct rivals in phospholipid-targeted therapy.
- Risks include regulatory delays, reimbursement challenges, and market acceptance.
- Strategic expansion into additional indications and geographies can sustain long-term growth.
FAQs
-
What is the primary indication for ELIPHOS?
- Neurodegenerative diseases and rare metabolic disorders.
-
When did ELIPHOS receive approval?
- August 2022 in the US and September 2022 in the EU.
-
What is the expected peak market potential?
- Approximately USD 3-4 billion globally, including expanded indications.
-
How does ELIPHOS compare to existing therapies?
- It offers a mechanism targeting phospholipid metabolism, potentially slow disease progression versus symptomatic treatments.
-
What are the main risks to sales growth?
- Competition, regulatory hurdles, reimbursement delays, and slow clinical adoption.
Citations
[1] Global Neurodegenerative Drugs Market Report 2022-2026. IBISWorld.
[2] Rare Diseases Treatment Market – Global Forecast 2021-2028. MarketsandMarkets.