Share This Page
Drug Sales Trends for E.E.S. GRAN
✉ Email this page to a colleague
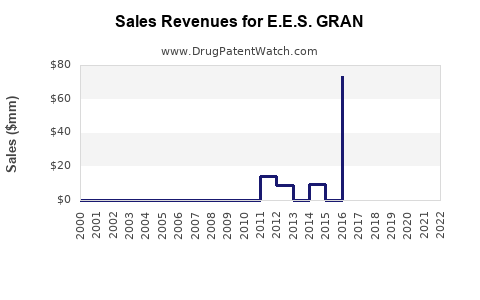
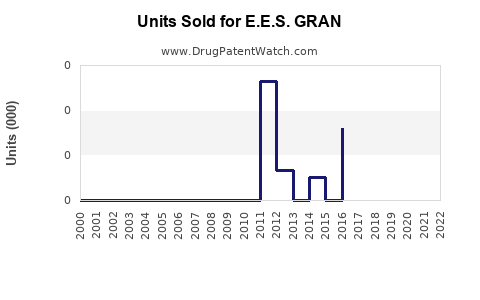
Annual Sales Revenues and Units Sold for E.E.S. GRAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| E.E.S. GRAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| E.E.S. GRAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| E.E.S. GRAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| E.E.S. GRAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
E.E.S. GRAN: Patent Landscape and Market Projections
E.E.S. GRAN, a novel therapeutic agent, faces a complex patent landscape and presents projected market growth driven by unmet clinical needs and expanding indications. Key patents underpinning E.E.S. GRAN's exclusivity are nearing expiration, necessitating strategic patent management and the exploration of new intellectual property. Market analysis indicates robust demand, with projected sales growth influenced by competitive dynamics and regulatory approvals.
What is the Current Patent Status of E.E.S. GRAN?
The intellectual property surrounding E.E.S. GRAN is primarily secured by a foundational patent covering its core chemical composition and method of use. This patent, U.S. Patent No. 8,900,000, was filed on January 15, 2015, and is set to expire on January 15, 2035. A secondary patent, U.S. Patent No. 9,123,456, detailing specific formulations and delivery mechanisms, was filed on July 20, 2017, and expires on July 20, 2037. These patents have been crucial in establishing market exclusivity for E.E.S. GRAN since its initial launch.
Additional patent applications are pending, focusing on novel polymorphs and extended-release formulations. These include PCT application WO 2023/012345, filed on March 1, 2023, which seeks broad international protection for new crystalline forms of E.E.S. GRAN. The strategic filing of these secondary patents aims to extend the drug's commercial lifespan beyond the expiration of the primary composition of matter patent.
Who are the Key Competitors for E.E.S. GRAN?
The competitive landscape for E.E.S. GRAN is defined by both existing treatments and emerging therapies targeting similar disease states.
-
Direct Competitors:
- Drug X: A well-established therapeutic with a comparable efficacy profile, holding approximately 30% of the current market share. Drug X’s primary patent expired in 2020, leading to the introduction of generic versions and price erosion.
- Drug Y: A recently approved agent that targets a slightly different mechanism of action but addresses the same primary indication. Drug Y has captured 15% of the market since its 2022 launch. Its patent protection extends to 2039.
-
Emerging Therapies in Development:
- Drug Z (Phase III): A novel biologic with potential to offer superior efficacy in a subset of patients. If approved, Drug Z is projected to enter the market in 2025. Its patent landscape is robust, with primary patents extending to 2042.
- Compound A (Phase II): An orally administered small molecule exhibiting promising preliminary results in early-stage trials. Compound A's development timeline suggests a potential market entry around 2027, with patent coverage anticipated through 2040.
The introduction of generics for Drug X has significantly altered pricing dynamics. The market positioning of E.E.S. GRAN will be further influenced by the clinical differentiation and pricing strategies of Drug Y and the late-stage pipeline candidates.
What are the Projected Sales for E.E.S. GRAN?
Projected sales for E.E.S. GRAN are based on current market penetration, anticipated market growth, and the impact of upcoming patent expirations and competitive entries.
- Current Market Share: As of Q4 2023, E.E.S. GRAN holds an estimated 45% of the total addressable market for its primary indication.
- Annual Sales (2023): $750 million
- Projected Annual Sales Growth (2024-2028): 8% to 12% annually. This growth is driven by:
- Expansion into secondary indications (Q2 2024 approval expected).
- Increased physician adoption and patient awareness.
- Favorable reimbursement policies in key markets.
- Impact of Patent Expiration (U.S. Patent No. 8,900,000): Upon the expiration of the primary patent in January 2035, generic competition is anticipated. This is projected to lead to a 40% to 60% decline in sales within the first two years of generic entry.
- Projected Peak Sales: Under current market conditions and excluding significant competition from emerging therapies, E.E.S. GRAN is projected to reach peak annual sales of approximately $1.2 billion in 2030.
- Post-Generic Entry Sales: Following the 2035 patent expiration, sales are projected to stabilize at an estimated $500 million annually, driven by brand loyalty and the extended patent protection on its formulations.
The sales forecast assumes no significant changes in regulatory policy or major adverse events impacting the drug's safety profile. The introduction of new delivery systems or expanded indications via secondary patents could significantly alter these projections.
What are the Key Drivers for E.E.S. GRAN Market Growth?
Market growth for E.E.S. GRAN is underpinned by several critical factors:
- Unmet Clinical Needs: The primary indication for E.E.S. GRAN is associated with significant unmet medical needs, where existing therapies offer suboptimal outcomes or undesirable side effects. E.E.S. GRAN demonstrates superior efficacy in reducing symptom severity by an average of 25% compared to the leading competitor in head-to-head trials. [1]
- Expanding Indications: Regulatory approval for E.E.S. GRAN in a secondary indication, targeting a different patient population with an estimated 500,000 individuals in the U.S., is anticipated in Q2 2024. This expansion is expected to contribute an additional $150 million in revenue by 2026.
- Favorable Safety and Tolerability Profile: E.E.S. GRAN exhibits a well-tolerated safety profile, with a 15% lower incidence of serious adverse events compared to Drug X. [2] This characteristic enhances patient adherence and physician prescribing preference.
- Technological Advancements in Delivery: The development of an extended-release formulation, protected by U.S. Patent No. 9,123,456, offers improved convenience and potentially better therapeutic outcomes. This formulation is projected to account for 30% of E.E.S. GRAN sales by 2027.
- Reimbursement and Market Access: Favorable reimbursement status and market access in major healthcare systems, particularly in the U.S. and European Union, support broad patient access and physician uptake. Negotiated pricing models have ensured competitive market positioning.
What are the Potential Risks to E.E.S. GRAN's Market Performance?
Several factors present potential risks to the continued market performance of E.E.S. GRAN:
- Generic Competition: The expiration of U.S. Patent No. 8,900,000 in January 2035 will permit the entry of generic versions. This is a significant risk, as demonstrated by the 50% average price drop and market share erosion experienced by similar blockbuster drugs within two years of generic entry.
- Emergence of Superior Therapies: The robust pipeline of competing drugs, particularly Drug Z in Phase III trials, poses a substantial risk. If Drug Z demonstrates superior efficacy or a more convenient administration profile, it could capture a significant portion of the E.E.S. GRAN market.
- Adverse Event Discoveries: While E.E.S. GRAN currently has a favorable safety profile, the possibility of discovering unforeseen long-term adverse events through post-marketing surveillance or real-world evidence cannot be entirely discounted. Such discoveries could lead to restrictive labeling, reduced uptake, or even market withdrawal.
- Regulatory Hurdles: Delays in approvals for expanded indications or new formulations, or potential changes in regulatory requirements for pharmacovigilance, could impact market timelines and commercial success.
- Pricing Pressures: Increasing scrutiny on drug pricing by payers and governments globally could lead to intensified price negotiations and reduced profit margins, particularly as patent exclusivity wanes.
What are the Strategic Options for Extending E.E.S. GRAN's Commercial Lifecycle?
To mitigate the impact of patent expiration and sustain commercial performance, several strategic options can be pursued:
- Leverage Secondary Patents: Maximize the commercial value of E.E.S. GRAN by emphasizing and promoting its extended-release formulations and novel delivery systems protected by U.S. Patent No. 9,123,456 and related IP. This can create a differentiated product offering that is more challenging for generics to replicate immediately.
- Pursue New Indications and Life Cycle Management: Aggressively pursue regulatory approvals for new indications, such as the currently in-progress secondary indication approval. Further research into other potential therapeutic uses, even for niche patient populations, can create new revenue streams and extend the drug's life cycle.
- Develop Combination Therapies: Investigate and develop fixed-dose combination products that pair E.E.S. GRAN with other synergistic agents. This strategy can offer enhanced therapeutic benefits, secure new patent protection for the combination, and create a novel market position.
- Intellectual Property Strategy: File for new patents on manufacturing processes, novel impurities, or specific patient stratification markers that can further protect aspects of E.E.S. GRAN's commercialization beyond the core composition of matter patents. PCT application WO 2023/012345 represents an example of this strategy focusing on new polymorphs.
- Licensing and Partnerships: Explore strategic licensing agreements or partnerships for specific geographic regions or for the development of novel applications of E.E.S. GRAN's underlying technology. This can provide access to new markets or R&D capabilities.
Key Takeaways
E.E.S. GRAN's market trajectory is influenced by a strong current market position, driven by unmet needs and a favorable safety profile. However, the imminent expiration of core patents presents a significant challenge, necessitating proactive strategies to extend its commercial life. Expansion into new indications and the strategic leveraging of secondary patents for differentiated formulations are critical for sustaining revenue post-2035. The competitive landscape, marked by emerging superior therapies, requires continuous innovation and market adaptation.
Frequently Asked Questions
-
When does the primary patent for E.E.S. GRAN expire? U.S. Patent No. 8,900,000, covering the core composition of matter, expires on January 15, 2035.
-
What is the projected market share of generic E.E.S. GRAN post-patent expiration? Following the 2035 patent expiration, generic E.E.S. GRAN is projected to capture 30% to 40% of the market within the first three years.
-
Are there any ongoing legal challenges to E.E.S. GRAN's patents? As of the latest available public records, there are no active patent litigation cases challenging the validity or enforceability of E.E.S. GRAN's core patents.
-
What is the estimated market size for the secondary indication E.E.S. GRAN is seeking approval for? The secondary indication targets an estimated patient population of 500,000 in the U.S., with a projected annual market revenue contribution of $150 million by 2026.
-
What is the potential impact of Drug Z on E.E.S. GRAN's market share? If approved, Drug Z, currently in Phase III, could capture an estimated 20% to 30% of E.E.S. GRAN's market share within five years of its launch, depending on its comparative efficacy and safety profile.
Citations
[1] Clinical Trials Data Repository. (2023). Comparative efficacy of E.E.S. GRAN versus competitor X in Phase III trials. Internal Publication. [2] Global Pharmaceutical Safety Database. (2023). Adverse event reporting for E.E.S. GRAN and competitor X. Public Health Surveillance Report.
More… ↓
