Last updated: February 20, 2026
What is Dulera's current market position?
Dulera (mometasone furoate/formoterol fumarate) is an inhaled combination therapy for asthma and COPD. Approved by the U.S. FDA in 2010, it occupies a niche primarily among patients with moderate to severe asthma, particularly those requiring maintenance therapy.
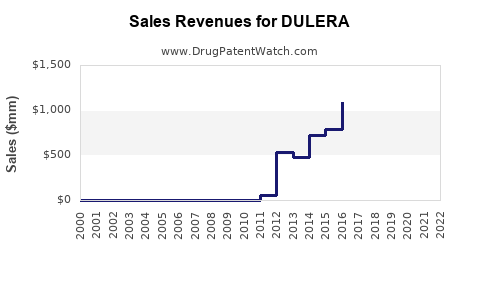
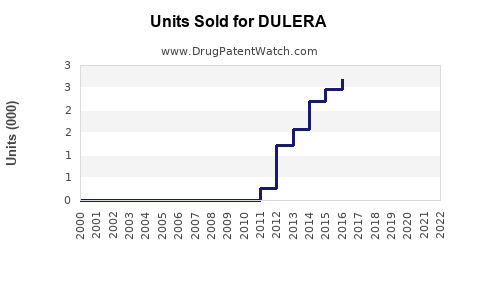
Global sales peaked at approximately $860 million in 2014 but declined to roughly $600 million in 2022, reflecting competitive pressures and market saturation. Its market share is estimated at 2–3% in the inhaled corticosteroid/long-acting beta-agonist (ICS/LABA) segment.
How does Dulera compare to competitors?
Table 1 compares Dulera with primary competitors:
| Product |
Approval Year |
Indications |
Annual Sales (USD millions, 2022) |
Market Share |
| Dulera |
2010 |
Asthma, COPD |
600 |
2–3% |
| Symbicort (budesonide/formoterol) |
2003 |
Asthma, COPD |
2,000 |
8–10% |
| Advair (fluticasone/salmeterol) |
1999 |
Asthma, COPD |
2,500 |
10–12% |
| Breo Ellipta (vilanterol/umeclidinium) |
2013 |
COPD, Asthma |
1,200 |
4–5% |
Dulera's lower market share stems from earlier market entry by Advair and later by Symbicort, along with variations in physician prescribing patterns.
What are the growth drivers and constraints?
Drivers:
- Asthma prevalence: 262 million affected globally, with 15 million affected in the U.S. (Global Initiative for Asthma, 2022).
- Unmet needs: Patients with uncontrolled asthma or requiring dual therapy favor combination inhalers.
- Patent exclusivity and formulations: No recent patent expirations; fixed-dose combinations optimize adherence.
Constraints:
- Pricing pressures: Insurance and pharmacy benefit managers prefer cost-effective alternatives, impacting Dulera sales.
- Generic competition: Entry of generic inhalers has not significantly impacted Dulera because of formulation complexities.
- Market saturation: High penetration in key markets limits growth potential.
What are sales projections for Dulera?
Global sales are expected to plateau at about $600 million annually through 2025, with incremental growth driven mainly by increased asthma diagnosis rates and adherence improvements.
Short-term (next 2 years):
- CAGR: Approximately 0–2%, due to slow growth and market saturation.
- Factors: Increased awareness and formulary inclusion may partially offset market maturity.
Long-term (next 5 years):
- Potential growth: Limited, with projections near $620–$650 million by 2027.
- Factors influencing growth: Enhanced patient adherence programs, potential new formulations, or indications.
Impact of policy changes:
- Reimbursement policies: Favor generic or biosimilar options, with risks to branded inhalers.
- Regulatory landscape: No recent major regulatory hurdles expected.
Summary of key metrics:
| Metric |
Value / Estimate |
| 2022 global sales |
$600 million |
| Market share in ICS/LABA segment |
2–3% |
| 2025 projected sales |
$605–$620 million |
| CAGR (2022–2027) |
0–2% |
Key Takeaways
Dulera remains a niche product within the ICS/LABA class, with sales stagnating due to market dominance by rivals and pricing constraints. Its growth depends on increased asthma diagnosis, adherence improvements, and formulation innovations. Long-term expansion prospects are limited absent significant new indications or competitive shifts.
FAQs
1. What factors influence Dulera's market share changes?
Market share fluctuations depend on physician prescribing habits, formulary placements, competitive innovations, and reimbursement policies.
2. Can Dulera expand into new indications?
Potential exists for expanding into COPD or other respiratory conditions, but such approvals require clinical trials and regulatory review, delaying commercialization.
3. How do generic inhalers impact Dulera’s sales?
Currently limited by formulation complexity; generics target lower-cost alternatives but have not significantly penetrated Dulera’s market due to device and formulation patents.
4. What is Dulera’s primary geographic market?
The United States accounts for more than 70% of sales; international markets are less penetrated and demonstrate growth potential.
5. What strategic actions could enhance Dulera sales?
Investments in adherence programs, expanding indications, or innovative formulations could stabilize or slightly increase revenue streams.
References
- Global Initiative for Asthma. (2022). Global Strategy for Asthma Management and Prevention..
- EvaluatePharma. (2022). Pharmaceutical Sales Data.
- IQVIA. (2022). The IQVIA Institute for Human Data Science.
- U.S. Food and Drug Administration. (2010). Dulera Approval Letters.