Share This Page
Drug Sales Trends for DOXYCYCL HYC
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for DOXYCYCL HYC (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
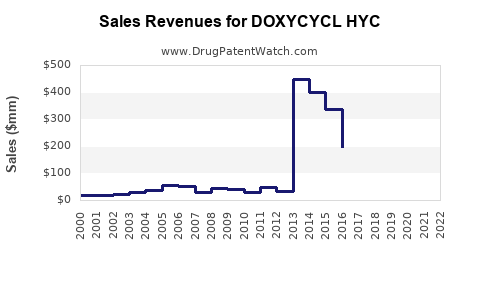
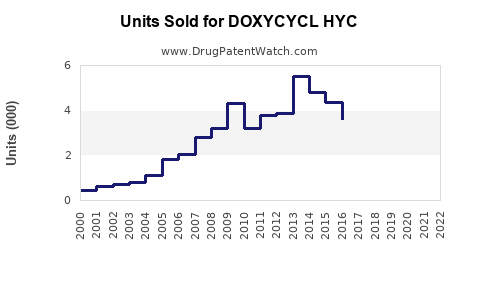
Annual Sales Revenues and Units Sold for DOXYCYCL HYC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DOXYCYCL HYC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DOXYCYCL HYC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DOXYCYCL HYC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Doxycycline Hydrate Market Analysis and Sales Projections
Doxycycline hydrate, a broad-spectrum tetracycline antibiotic, demonstrates robust market performance driven by its established efficacy in treating a wide range of bacterial infections and its utilization in acne management. The global market is projected to experience sustained growth through 2030, supported by increasing prevalence of infectious diseases, growing awareness of antibiotic resistance management, and continued demand in dermatological applications. Key market segments include infectious disease treatment, acne therapy, and potential off-label uses.
What is the current market size and projected growth for Doxycycline Hydrate?
The global doxycycline hydrate market is a significant segment within the broader antibiotic market. In 2023, the market was valued at approximately $1.2 billion. Projections indicate a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, estimating the market to reach $1.7 billion by the end of the forecast period. This growth is underpinned by several factors, including the persistent need for broad-spectrum antibiotics, particularly in developing economies where infectious diseases remain a public health concern. Furthermore, the expanding application of doxycycline in managing inflammatory conditions, such as rosacea and acne vulgaris, contributes to market expansion. The increasing incidence of sexually transmitted infections (STIs) also bolsters demand for doxycycline, a first-line treatment for conditions like chlamydia and gonorrhea.
What are the primary therapeutic indications for Doxycycline Hydrate?
Doxycycline hydrate is indicated for the treatment of a diverse array of bacterial infections. Its primary therapeutic applications include:
- Respiratory Tract Infections: Effective against Streptococcus pneumoniae, Haemophilus influenzae, and Mycoplasma pneumoniae, it is used for conditions such as pneumonia, bronchitis, and sinusitis.
- Urinary Tract Infections (UTIs): Treats UTIs caused by susceptible Gram-positive and Gram-negative bacteria.
- Skin and Soft Tissue Infections: Addresses infections caused by Staphylococcus aureus and Streptococcus pyogenes, including cellulitis and impetigo.
- Sexually Transmitted Infections (STIs): A critical treatment for Chlamydia trachomatis and Neisseria gonorrhoeae (in specific regimens), and syphilis.
- Tick-Borne Diseases: Plays a vital role in treating Lyme disease (Borrelia burgdorferi), Rocky Mountain spotted fever (Rickettsia rickettsii), and ehrlichiosis.
- Acne Vulgaris: Widely prescribed for its anti-inflammatory and antibacterial properties to manage moderate to severe acne.
- Ophthalmic Infections: Used to treat conjunctivitis caused by susceptible bacteria.
- Peri-odontal Infections: Employed in the treatment of periodontitis.
- Malaria Prophylaxis: Utilized as a preventative measure against malaria in travelers to endemic areas.
The broad spectrum of activity and favorable pharmacokinetic profile contribute to its extensive use across these indications.
Which geographic regions represent the largest and fastest-growing markets for Doxycycline Hydrate?
The North American market, led by the United States, currently holds the largest share of the doxycycline hydrate market, accounting for approximately 35% of global sales. This dominance is attributed to high healthcare expenditure, advanced healthcare infrastructure, and a high prevalence of infectious diseases and dermatological conditions.
The Asia-Pacific region is projected to exhibit the fastest growth rate, with an estimated CAGR of 5.8% over the forecast period. This expansion is driven by increasing healthcare access, a large population base, a rising incidence of infectious diseases due to varying sanitation standards, and growing awareness and diagnosis of STIs. Key emerging markets within this region include China and India.
Europe represents the second-largest market, with steady growth anticipated due to an aging population susceptible to infections and established healthcare systems. Latin America and the Middle East & Africa are also expected to contribute to market growth, albeit at a more moderate pace, driven by improving healthcare infrastructure and increasing disease burden.
What are the key drivers and challenges impacting the Doxycycline Hydrate market?
Key Market Drivers:
- Increasing Prevalence of Infectious Diseases: Global outbreaks and endemic infectious diseases continue to necessitate broad-spectrum antibiotics like doxycycline. The rise in bacterial pneumonia, UTIs, and STIs directly fuels demand.
- Dermatological Applications: The persistent global prevalence of acne vulgaris and rosacea, coupled with doxycycline's efficacy as an anti-inflammatory and antibacterial agent, sustains strong demand from the dermatology segment.
- Antibiotic Resistance Management: While antibiotic resistance is a challenge, doxycycline’s established safety profile and efficacy against certain resistant strains, when used appropriately, maintain its therapeutic relevance. It is often used in combination therapies or as an alternative where other antibiotics are ineffective.
- Cost-Effectiveness: As a generic medication, doxycycline hydrate offers a cost-effective treatment option compared to newer, branded antibiotics, making it accessible in both developed and developing economies.
- Government Initiatives and Public Health Programs: Increased focus on combating STIs and other infectious diseases through public health campaigns and accessible treatment programs, particularly in low- and middle-income countries, supports market growth.
Key Market Challenges:
- Growing Antibiotic Resistance: The development of resistance by bacteria to doxycycline, particularly Neisseria gonorrhoeae, poses a significant challenge. This necessitates careful prescribing practices and the development of new treatment strategies.
- Competition from Newer Antibiotics: The introduction of novel antibiotics with broader spectra of activity or improved pharmacokinetic profiles can erode market share for older drugs like doxycycline.
- Stringent Regulatory Scrutiny: Regulatory bodies continuously review antibiotic usage due to resistance concerns, which can impact approval processes for new formulations or indications and lead to stricter prescribing guidelines.
- Side Effects and Adverse Reactions: Like all antibiotics, doxycycline can cause side effects, including gastrointestinal disturbances, photosensitivity, and tooth discoloration in children under eight, which can limit its use in certain patient populations.
- Availability of Counterfeit Products: The generic nature of doxycycline makes it susceptible to counterfeit products, particularly in unregulated markets, posing risks to patient safety and undermining legitimate manufacturers.
What is the competitive landscape for Doxycycline Hydrate?
The doxycycline hydrate market is highly fragmented and competitive, characterized by the presence of numerous generic manufacturers. Major pharmaceutical companies and a significant number of smaller generic drug producers compete based on price, product quality, and supply chain reliability.
Key players in the doxycycline hydrate market include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc.
- Hikma Pharmaceuticals PLC
- Aurobindo Pharma Limited
- Lupin Limited
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Bayer AG (though primarily focused on branded products, maintains a presence)
- Cipla Ltd.
- Gland Pharma Ltd.
The competitive strategy often revolves around optimizing manufacturing costs to offer competitive pricing, ensuring consistent supply chains, and adhering to stringent quality control measures to meet global regulatory standards. Companies also focus on obtaining and maintaining regulatory approvals in various key markets.
What are the future market trends and opportunities for Doxycycline Hydrate?
Future market trends for doxycycline hydrate are likely to be shaped by advancements in drug delivery, evolving resistance patterns, and expanding therapeutic niches.
- Development of Novel Formulations: Research into extended-release formulations or improved delivery systems could enhance patient compliance and potentially reduce the frequency of dosing, offering a competitive edge.
- Combination Therapies: Exploring the efficacy of doxycycline in combination with other agents to combat resistant bacterial strains or to enhance therapeutic outcomes in complex infections presents a significant opportunity.
- Repurposing and Off-Label Use Expansion: Further research into the anti-inflammatory and immunomodulatory properties of doxycycline may lead to expanded approved indications beyond its current antimicrobial uses, particularly in chronic inflammatory diseases.
- Focus on Emerging Markets: Continued penetration into emerging markets in Asia-Pacific, Latin America, and Africa, where infectious diseases and limited access to newer antibiotics persist, offers substantial growth potential.
- Addressing Resistance Through Stewardship: Manufacturers and healthcare providers will increasingly focus on antibiotic stewardship programs to ensure responsible use of doxycycline, thereby preserving its efficacy and extending its therapeutic lifespan. This includes developing educational materials and diagnostic tools that aid in appropriate prescribing.
Key Takeaways
The global doxycycline hydrate market is projected to grow from $1.2 billion in 2023 to $1.7 billion by 2030, driven by its broad-spectrum antibiotic activity, established efficacy in treating various infections and acne, and cost-effectiveness. North America currently leads the market, while the Asia-Pacific region is expected to witness the fastest growth. Key challenges include rising antibiotic resistance and competition from newer drugs. The market remains highly competitive with numerous generic manufacturers. Future opportunities lie in novel formulations, combination therapies, and expanding applications in emerging markets.
Frequently Asked Questions
How does antibiotic resistance affect the future demand for Doxycycline Hydrate?
Antibiotic resistance to doxycycline, particularly in pathogens like Neisseria gonorrhoeae, necessitates a careful approach to its use. While resistance can reduce demand for monotherapy in certain infections, doxycycline remains a valuable agent in combination therapies and for infections where resistance is not yet widespread or for patients intolerant to other antibiotics. Responsible antibiotic stewardship programs aim to preserve the efficacy of doxycycline, ensuring its continued utility.
What regulatory hurdles might Doxycycline Hydrate manufacturers face?
Manufacturers face ongoing regulatory scrutiny regarding product quality, manufacturing practices (cGMP), and post-market surveillance. Concerns over antibiotic resistance may lead to stricter prescribing guidelines or requirements for specific indications. Obtaining and maintaining marketing authorizations in diverse global markets requires adherence to varied national regulatory standards.
Are there significant R&D investments in new doxycycline hydrate-based drugs?
Given that doxycycline hydrate is a well-established generic drug, large-scale R&D investments are typically focused on optimizing manufacturing processes, developing novel drug delivery systems (e.g., extended-release formulations), or exploring its potential in combination therapies rather than developing entirely new molecular entities based on doxycycline. Research into repurposing its anti-inflammatory properties is also an area of interest.
How does the pricing strategy of generic doxycycline hydrate impact market dynamics?
The low cost of generic doxycycline hydrate is a primary driver of its widespread use, particularly in resource-limited settings. This pricing strategy makes it accessible for treating common infections and acne. Intense price competition among generic manufacturers is a hallmark of this market segment, with companies focusing on cost efficiencies in production and supply chain management.
What is the role of Doxycycline Hydrate in treating non-infectious inflammatory conditions?
Beyond its antimicrobial properties, doxycycline hydrate exhibits anti-inflammatory effects. This has led to its use in treating non-infectious inflammatory conditions such as rosacea and periodontitis. Research is ongoing to further understand and potentially expand its therapeutic applications in other inflammatory diseases, representing a potential growth avenue for the market.
Citations
[1] Global Market Insights. (2023). Doxycycline Market Size, Share & Industry Analysis. [2] Grand View Research. (2023). Doxycycline Market Size, Share & Trends Analysis Report. [3] Allied Market Research. (2023). Doxycycline Market by Product Type, Formulation, and End-Use: Global Opportunity Analysis and Industry Forecast, 2023-2032. [4] Mordor Intelligence. (2023). Doxycycline Market - Growth, Trends, COVID-19 Impact, and Forecasts. [5] U.S. Food and Drug Administration. (n.d.). Doxycycline. Retrieved from fda.gov
More… ↓
