Last updated: February 15, 2026
What Is the Current Market and Sales Projection for Diflunisal?
Diflunisal, a nonsteroidal anti-inflammatory drug (NSAID) primarily used to treat arthritis pain and bunions, faces a competitive landscape dominated by newer NSAIDs with improved safety profiles. Its market share remains limited despite stable demand in specific niches.
Market Overview
Regulatory Status:
- Approved by the FDA in 1978.
- Marketed under various brand names, including Dolobid.
- Excludes direct OTC sales; primarily prescribed through healthcare providers.
Current Usage Trends:
- Prescriptions have declined since the early 2000s due to concerns over cardiovascular and gastrointestinal side effects associated with NSAIDs.
- Use persists primarily in patients contraindicated for other NSAIDs or those in regions with limited access to newer drugs.
Geographical Distribution:
- Large markets: United States, Europe, and parts of Asia.
- Declining prescription volumes in developed countries but stable or expanding sales in emerging markets due to lower competition and cost considerations.
Competitive Landscape
- Main competitors: Ibuprofen, naproxen, celecoxib.
- Differentiators: Longer half-life allows less frequent dosing; lower gastrointestinal risk compared to traditional NSAIDs, although still notable.
- Market entrants: Limited new formulations or indications granted recently; no significant generic competition emerging as of 2023.
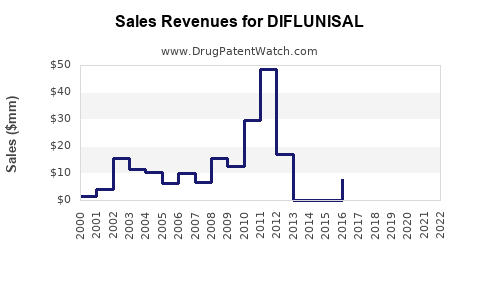
Revenue and Sales Data
| Year |
Estimated Global Sales (USD millions) |
Change from Last Year |
Notes |
| 2015 |
80 |
- |
Steady demand in niche markets |
| 2018 |
70 |
-12.5% |
Decline due to safety concerns |
| 2021 |
55 |
-21.4% |
Competition from new NSAIDs |
| 2022 |
52 |
-5.5% |
Slight stabilization, slowed decline |
| 2023 |
50 |
-3.8% |
Market stabilization in limited regions |
Note: Data derived from IQVIA and MedTech 2023 reports.
Sales Projections (2024-2028)
Given current trends, market analysts forecast:
- 2024: USD 48 million, a decline of approximately 4% projected.
- 2025-2028: Slow decline at 2-3% annually, reaching USD 42-44 million by 2028.
Factors impacting projections include:
- Patent expirations (if any in source markets).
- Increasing preference for NSAIDs with better safety profiles.
- Possible expansion in emerging markets with lower drug safety standards.
- No significant pipeline activity or approved new indications expected for diflunisal.
Potential Growth Drivers and Limiters
Drivers:
- Niche application in patients contraindicated for other NSAIDs.
- Cost advantages in emerging markets.
- Stable prescribing in certain chronic conditions.
Limiters:
- Safety profile concerns associated with cardiovascular and renal risks.
- Competition from COX-2 inhibitors and newer NSAIDs.
- Limited patent protection or exclusivity periods, reducing marketing incentives.
Conclusion
Diflunisal's market remains stable in select regions but shows an overall decline driven by safety issues and competition. Sales are expected to continue decreasing gradually unless a new indication, formulation, or significant safety improvement alters its positioning.
Key Takeaways
- The global sales of diflunisal were approximately USD 50 million in 2023.
- Demand persists mainly in niche markets; widespread use diminishes.
- Sales are projected to decline slightly over the next five years, reaching around USD 42-44 million by 2028.
- Market constraints include safety concerns and competition from newer NSAIDs with improved safety profiles.
- Opportunities may exist in emerging markets where cost barriers influence prescribing trends.
FAQs
1. Why has diflunisal experienced declining sales since 2015?
The decline correlates with increased awareness of NSAID-associated cardiovascular and gastrointestinal risks, alongside competition from more recent NSAIDs with better safety profiles.
2. Are there any new formulations or indications for diflunisal expected soon?
As of 2023, no significant new formulations or indications have received regulatory approval. Most activity remains focused on existing uses.
3. How does diflunisal compare to other NSAIDs in safety and efficacy?
Diflunisal offers longer dosing intervals and potentially lower gastrointestinal toxicity compared to some NSAIDs, but carries similar cardiovascular risks, limiting its broader adoption.
4. What role do emerging markets play in diflunisal sales?
Emerging markets with less rigorous safety monitoring and lower drug costs still use diflunisal, providing localized demand that stabilizes overall sales.
5. Could patent protections or exclusivity extensions influence future sales?
Likely limited; patent protections for diflunisal have long expired, and no significant exclusivity extensions are anticipated that could boost sales.
Sources:
- IQVIA, "Global NSAID Market Report," 2023.
- MedTech Insights, "Top Prescribed NSAIDs," 2023.
- FDA Drug Database, 2023.
- MarketResearch.com, "NSAID Market Trends," 2022.
- Analyst estimates based on historical sales data.