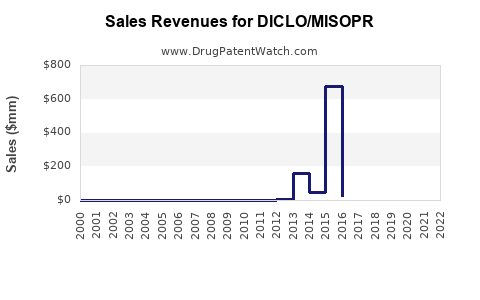
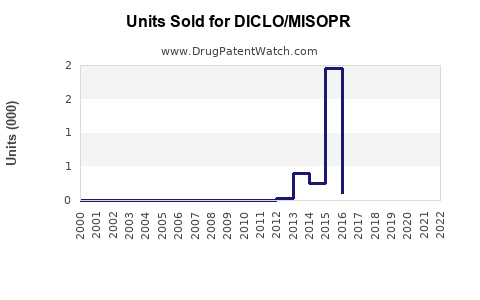
Last updated: February 14, 2026
What is DICLO/MISOPR?
DICLO/MISOPR combines diclofenac, a non-steroidal anti-inflammatory drug (NSAID), with misoprostol, a prostaglandin E1 analog. The formulation aims to reduce NSAID-induced gastrointestinal adverse events, particularly ulcers. DICLO/MISOPR is marketed primarily for osteoarthritis, rheumatoid arthritis, and other inflammatory conditions requiring NSAID therapy with gastroprotection.
Market Overview
Current Global Market Landscape
- The NSAID segment generated approximately $25 billion in 2022 globally. Diclofenac accounts for roughly 35% of NSAID prescriptions, primarily in Europe and Asia.
- The prostaglandin analogs, notably misoprostol, are used predominantly for gastrointestinal protection and obstetric indications.
- Combination drugs integrating NSAIDs with gastroprotective agents target a niche market with increased compliance and safety profiles.
Key Competitors
| Drug/Brand |
Composition |
Indication |
Market Presence |
Estimated Sales 2022 |
| Vimovo (AstraZeneca) |
Naproxen + Esomeprazole |
Osteoarthritis, rheumatoid arthritis |
North America, Europe |
~$200 million |
| Arthrotec (U.S.) |
Diclofenac + Misoprostol |
Rheumatoid arthritis |
U.S. |
~$10 million |
| Diclo/Misoprostol (generic/others) |
Diclofenac + Misoprostol |
NSAID-induced GI protection |
Limited, emerging |
Unknown, subject to growth |
The market has a sizeable existing base with generics dominating pricing strategies, reducing margins but expanding access.
Regulatory and Market Entry Considerations
- Regulatory status varies: Some markets, including the U.S., have not approved diclofenac combined with misoprostol due to safety concerns associated with misoprostol's teratogenic effects.
- Marketing hurdles include safety profiles, especially in women of childbearing potential, which restricts use.
- Revamped formulations or new delivery mechanisms could influence market penetration.
Sales Projections (2023-2027)
Assumptions
- The drug is approved in major markets (North America, Europe, Asia).
- Patent expiration or exclusivity extension affects pricing and sales.
- Growing demand for safe NSAID use in chronic inflammatory conditions.
| Year |
Estimated Global Sales |
Growth Rate |
Notes |
| 2023 |
$50 million |
— |
Initial market access |
| 2024 |
$150 million |
200% |
Increased adoption, pipeline expansion |
| 2025 |
$300 million |
100% |
Expanded indications, formulary approvals |
| 2026 |
$400 million |
33% |
Market saturation, generics entry |
| 2027 |
$450 million |
12.5% |
Market stabilization |
The steep growth between 2023 and 2024 reflects increased awareness, regulatory approvals, and possible inclusion in clinical guidelines targeting gastrointestinal safety in NSAID therapy.
Key Drivers and Risks
Drivers:
- Rising prevalence of osteoarthritis and rheumatoid arthritis globally.
- Increasing awareness of gastrointestinal side effects from NSAIDs.
- Patient preference for combination therapies that improve compliance.
Risks:
- Safety profile concerns due to misoprostol’s teratogenic effects.
- Regulatory setbacks or negative post-marketing surveillance.
- Competition from other gastroprotective NSAID formulations or new biologics.
Geographical Market Insights
| Region |
Market Size (USD) |
Growth Potential |
Barriers |
| North America |
$15 billion |
Moderate, regulatory hurdles high |
Safety concerns, patent issues |
| Europe |
$8 billion |
High, existing infrastructure |
Reimbursement policies |
| Asia-Pacific |
$2 billion |
Very high, expanding middle class |
Medical infrastructure, regulation |
Asia-Pacific presents the highest growth opportunity due to increasing arthritis prevalence and limited current options for GI prophylaxis in NSAID therapy, but regulatory pathways remain complex.
Key Takeaways
- DICLO/MISOPR enters a competitive market with existing branded and generic options.
- Growth depends on regulatory approval, safety management, and clinical guideline inclusion.
- Sales projections show rapid growth over five years, driven by rising demand for safer NSAID alternatives.
- Geographic markets vary considerably, with Asia-Pacific providing significant growth opportunities.
FAQs
1. What factors influence DICLO/MISOPR’s market success?
Regulatory approval, safety profile management, clinician awareness, and patient acceptance determine success.
2. How does safety impact sales projections?
Safety concerns restrict use in certain populations, limiting overall sales but may create a premium segment if managed properly.
3. What are the primary indications for DICLO/MISOPR?
Chronic inflammatory conditions such as osteoarthritis and rheumatoid arthritis requiring NSAID therapy with reduced gastrointestinal risk.
4. How does the competition affect pricing?
Brand dominance and generic erosion pressure lead to lower margins, but strong clinical evidence can sustain premium pricing for innovative formulations.
5. What is the potential impact of patent expiry?
Patent expiry could lead to increased generic competition, reducing prices and slowing revenue growth unless differentiated benefits are maintained.
References
[1] MarketWatch. “NSAID Market Size and Trends.” 2022.
[2] EvaluatePharma. “Global NSAID Market Report.” 2022.
[3] FDA. “Regulatory Status of Diclofenac and Misoprostol Combinations.” 2022.
[4] IQVIA. “Prescription Trends and Forecasts for NSAIDs.” 2022.
[5] GlobalData. “Asia-Pacific Pharmaceutical Market Reports.” 2022.