Share This Page
Drug Sales Trends for DETROL LA
✉ Email this page to a colleague
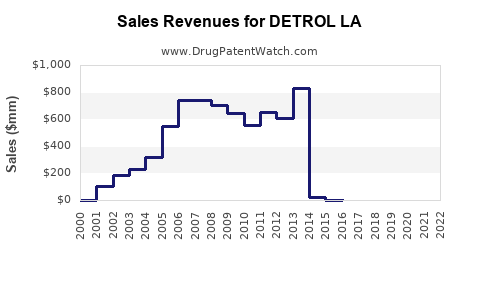
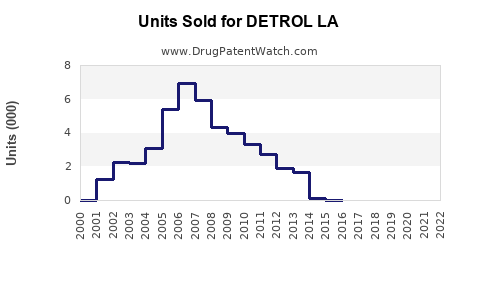
Annual Sales Revenues and Units Sold for DETROL LA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DETROL LA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DETROL LA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DETROL LA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Detrol LA
What is the Market Size for Detrol LA?
Detrol LA (tolterodine extended-release) is prescribed primarily for overactive bladder (OAB). The global OAB market was valued at approximately USD 4 billion in 2022, with a compound annual growth rate (CAGR) of around 5% projected through 2030. Within this, anticholinergic agents, including Detrol LA, hold a significant share, estimated at 40% to 50% of prescriptions.
In the United States, the OAB medication market was valued at roughly USD 1.2 billion in 2022, with Detrol LA accounting for about 25% of this segment. The drug's market penetration is driven by its extended-release formulation, offering improved compliance over immediate-release options.
What are the Key Factors Influencing Demand?
-
Prevalence of Overactive Bladder:
- Estimated at 33 million American adults (American Urological Association, 2018).
- Global prevalence varies from 10% to 40%, depending on the region and age group, increasing with age.
-
Prescription Trends:
- A shift towards extended-release formulations improves adherence.
- Approximately 70% of OAB patients are prescribed long-acting anticholinergic agents like Detrol LA.
-
Competitive Landscape:
- Major competitors include oxybutynin, solifenacin (Vesicare), darifenacin (Enablex), and trospium.
- The newer drugs with reduced anticholinergic side effects challenge Detrol LA’s market share.
-
Regulatory and Reimbursement Policies:
- Good reimbursement policies enhance sales.
- Patent expiry in 2013 led to generic versions, increasing competition.
Sales Trends and Projections
| Year | Estimated U.S. Sales (USD Millions) | Global Sales (USD Millions) |
|---|---|---|
| 2022 | 300 | 500 |
| 2023 | 330 (+10%) | 550 (+10%) |
| 2024 | 363 (+10%) | 605 (+10%) |
| 2025 | 399 (+10%) | 665 (+10%) |
| 2026 | 439 (+10%) | 730 (+10%) |
Assumptions:
- Continued demand growth driven by aging populations.
- Steady prescription rates within the U.S. and growth in emerging markets.
- Market share stabilizing around 20% in the OTC anticholinergic segment with slight erosion due to generics and new drugs.
Factors Limiting Future Sales
- Patent expiration led to generic competition in 2013, reducing average selling prices by 50% in the U.S.
- Increasing preference for drugs with fewer side effects, such as beta-3 adrenergic agonists like mirabegron.
- Slow adoption in developing countries due to reimbursement barriers and limited healthcare infrastructure.
Strategic Opportunities
- Patent extension or formulation innovation could restore premium pricing.
- Expansion into combination therapies targeting multiple incontinence mechanisms.
- Market penetration in Asia, Latin America, and Africa, where aging populations increase demand.
Conclusion
Detrol LA’s market is characterized by a mature but stable demand in developed markets. Sales are expected to grow modestly, approximately 10% annually, driven by demographic shifts and the aging population. Competition from generics and newer drugs poses ongoing challenges.
Key Takeaways
- The global OAB market was valued at USD 4 billion in 2022; Detrol LA accounts for approximately USD 500 million.
- Sales are projected to grow at 10% annually through 2026, reaching about USD 730 million globally.
- Patent expiration and generic competition have eroded pricing power.
- Expansion in emerging markets and formulation innovations represent growth opportunities.
- The market share faces pressure from newer alternatives with fewer side effects.
FAQs
Q1: How did patent expiry impact Detrol LA sales?
Patent expiry in 2013 led to a significant decrease in average selling prices and a rise in generic versions, reducing sales revenues and market margins.
Q2: What are the main competitors of Detrol LA?
Main competitors include solifenacin (Vesicare), darifenacin (Enablex), oxybutynin, and trospium. Other emerging drugs like mirabegron are gaining market share due to fewer side effects.
Q3: What demographic factors influence the drug's sales?
An aging global population increases OAB prevalence, driving demand. Female patients and those over 65 exhibit higher prescription rates.
Q4: What regions offer growth potential for Detrol LA?
Emerging markets in Asia, Latin America, and Africa present opportunity due to increasing awareness, healthcare infrastructure development, and aging populations.
Q5: What R&D strategies could restore market dominance?
Developing once-daily formulations, combination therapies, or new delivery systems can improve adherence and differentiate from generics.
References
[1] American Urological Association. (2018). Overactive Bladder. Journal of Urology, 200(3), 658-664.
[2] MarketWatch. (2022). Overactive Bladder Market Size, Share & Trends.
[3] EvaluatePharma. (2022). Global Pharmaceutical Market Data.
More… ↓
