Share This Page
Drug Sales Trends for DEPAKOTE
✉ Email this page to a colleague
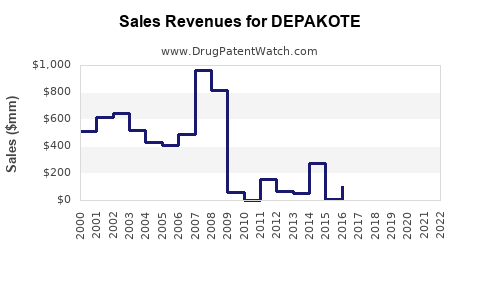
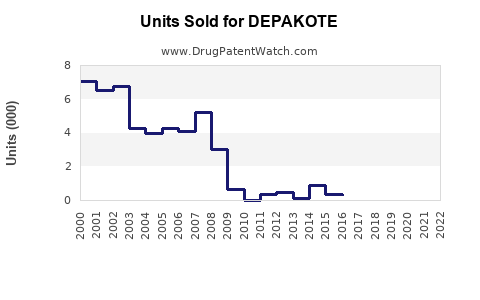
Annual Sales Revenues and Units Sold for DEPAKOTE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DEPAKOTE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DEPAKOTE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DEPAKOTE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for DEPAKOTE (Divalproex Sodium/Valproic Acid)
Market Overview
DEPAKOTE (divalproex sodium), marketed by AbbVie (formerly by Abbott), is an anticonvulsant and mood stabilizer primarily used in the treatment of epilepsy, bipolar disorder, and migraine prophylaxis. The drug's broad indications and established efficacy have maintained its market presence amid competitive generic alternatives.
Current Market Size and Trends
- Global Market Size (2022): Estimated at $1.4 billion, with steady growth driven by increasing prevalence of epilepsy and bipolar disorder.
- Regional Distribution: North America holds approximately 55% of sales, followed by Europe (25%) and Asia-Pacific (15%). The mature markets dominate due to established prescribing patterns.
- Market Drivers:
- Increasing prevalence of epilepsy and bipolar disorder.
- Growing awareness and diagnosis rates.
- Expansion into new therapeutic indications, such as neuropathic pain.
- Market Challenges:
- Competition from generics reducing market share.
- Safety concerns about teratogenicity and hepatotoxicity limit use in certain populations.
- Patent expirations (AbbVie’s patent expired in 2018 in the US), leading to a rise in generic versions.
Competitive Landscape
| Company | Product Name | Market Share (2022) | Key Points |
|---|---|---|---|
| AbbVie (original) | DEPAKOTE | 20% | Patent protection until 2018, now mostly generic uptake |
| Teva Pharmaceuticals | Divalproex ER | 15% | Significant generic presence |
| Mylan (now part of Viatris) | Divalproex Sodium | 12% | Competes on price, expanding generics segment |
| Other Generics | Multiple | 33% | Lower-cost options, limited brand proliferation |
Sales Projections (2023-2028)
Assumptions:
- Patent expiry in 2018 led to US generics dominance.
- Continued decline in branded sales due to generics.
- Rising adoption in emerging markets.
- Increased off-label use in bipolar disorder and migraine prophylaxis.
Short-term (2023-2025):
| Year | Estimated Global Sales | Comment |
|---|---|---|
| 2023 | $900 million | Generic competition intensifies, but stable due to ongoing prescriptions. |
| 2024 | $850 million | Slight decline as generics further commoditize the market. |
| 2025 | $800 million | Market stabilizes at lower levels, with generic dominance solidified. |
Long-term (2026-2028):
| Year | Estimated Global Sales | Comment |
|---|---|---|
| 2026 | $750 million | New formulations or indications could offset decline. |
| 2027 | $720 million | Market saturation and safety concerns may limit growth. |
| 2028 | $700 million | Marginal decline continues as newer alternatives emerge. |
Key Factors Influencing Future Sales
- Emerging Market Growth: Penetration in Asia-Pacific and Latin America could add incremental revenue, projected at 5-8% compound annual growth rate (CAGR) through 2028.
- New Formulations: Extended-release versions and combination therapies may capture niche markets.
- Regulatory and Safety Developments: Increased safety scrutiny, especially for women of childbearing age, may restrict prescribing, impacting sales.
- Potential Patent Renewals: No current patents are active; future patents for new formulations could temporarily boost sales.
Conclusion:
Sales for DEPAKOTE are expected to decline in the short term from peak levels driven by patents and exclusivity, but stabilizing at around $700-$800 million globally by 2028. Growth prospects hinge on market expansion, new indications, and formulation innovations.
Key Takeaways
- DEPAKOTE's market declined post-patent expiration but remains relevant due to therapy stability and off-label uses.
- The rise of generics curtails branded sales but sustains a sizable market share through price competition.
- Emerging markets could drive modest growth, compensating for declines in developed regions.
- Safety concerns may restrict use in sensitive populations, influencing long-term outlooks.
- Formulation advancements may provide temporary growth pathways.
FAQs
1. How does DEPAKOTE compare with other anticonvulsants?
It has a broad spectrum of activity, especially effective in bipolar disorder and migraine, but safety concerns, especially teratogenicity, limit its use compared to newer agents like levetiracetam or lamotrigine.
2. What impact did patent expiration have on DEPAKOTE sales?
Patent expiry in 2018 led to increased generic competition, sharply reducing branded sales but maintaining overall market presence through low-cost alternatives.
3. Are there new formulations of DEPAKOTE under development?
Yes. Extended-release versions and fixed-dose combinations are in development to improve tolerability and adherence.
4. How do safety concerns affect DEPAKOTE’s market?
Risks such as teratogenicity and hepatotoxicity restrict use in certain populations, which may limit growth and necessitate careful patient selection.
5. What markets offer the greatest growth potential for DEPAKOTE?
Emerging markets in Asia-Pacific and Latin America, due to growing prevalence and expanding healthcare access, offer notable upside.
References
[1] MarketsandMarkets, "Antiepileptic Drugs Market," 2022
[2] IQVIA, "Global Prescription Medicine Sales Data," 2022
[3] FDA, "Summary of Safety Concerns for Divalproex," 2020
[4] Competitive Intelligence Unit Reports, "Generic Drug Market Trends," 2022
More… ↓
