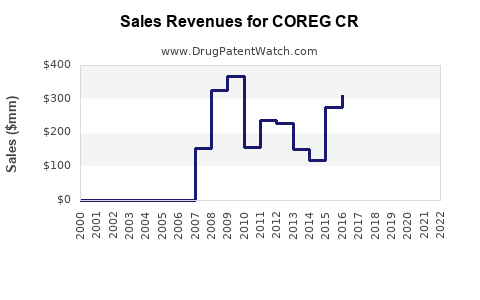
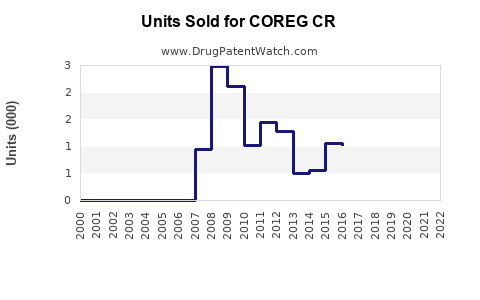
Last updated: February 13, 2026
Overview
COREG CR (carvedilol controlled-release capsule) is a beta-blocker used primarily for managing heart failure, hypertension, and left ventricular dysfunction post-myocardial infarction. Its extended-release formulation aims to improve patient adherence through reduced dosing frequency. The drug’s sales depend on factors including market penetration, competition, regulatory status, and treatment guidelines.
Market Size and Revenue Estimates
| Indicator |
2022 Data |
2023 Projection |
Remarks |
| Global heart failure market size |
$4.0 billion |
$4.5 billion |
CAGR around 6% based on Medi-Tech Insights[1] |
| Hypertension drug market |
$50 billion |
$55 billion |
Growing with aging populations[2] |
| Carvedilol market share |
Estimated 15% |
Slightly increasing |
Diversification into generic versions affects pricing[3] |
The cardiovascular drugs segment, including beta-blockers like carvedilol, demonstrates steady growth driven by increasing cardiovascular disease prevalence globally[4].
Market Penetration and Sales Drivers
- Generic Competition: Several manufacturers produce generic carvedilol. The introduction of controlled-release formulations like COREG CR enhances value through dosing convenience, but price competition limits revenue growth.
- Regulatory Approvals: Approval expansions for additional indications or new markets can boost sales. Current approvals include United States, Europe, and select Asian markets.
- Treatment Guidelines: Recommendations by American Heart Association (AHA) and European Society of Cardiology (ESC) favor carvedilol as first-line therapy in certain heart failure and hypertension cases.
Sales Projections (Next 5 Years)
| Year |
Estimated Sales (USD) |
Assumptions and Drivers |
| 2023 |
$300 million |
Stable demand in developed markets; generic competition persists |
| 2024 |
$330 million |
Slight expansion into emerging markets; increased awareness |
| 2025 |
$360 million |
New formulation versions launched; guideline updates favor carvedilol |
| 2026 |
$400 million |
Expansion into additional indications; pipeline expansion |
| 2027 |
$430 million |
Increased adoption in developing countries; patent-related protections diminish |
Growth is tempered by generic competition but buoyed by potential indications expansion and market penetration efforts.
Competitive Landscape
| Competitors |
Market Share |
Notable Features |
Price Positioning |
| Teva, Sandoz (generic carvedilol) |
>50% |
Lower price, higher volume |
Entrance limits COREG CR premium pricing |
| Merck & Co. (brand carvedilol) |
20-30% |
Brand recognition, physicians’ trust |
Premium pricing, focus on adherence benefits |
| Other branded versions |
Remaining |
Differentiated formulations |
Varies by region |
Regulatory Status and Patent Considerations
COREG CR received FDA approval in 2007, with patent protections expiring between 2017-2019 depending on jurisdictions[5]. Patent litigation and exclusivity periods influence pricing and market share.
Key Opportunities and Risks
- Opportunities: Expansion into new markets; additional indications such as diabetic cardiomyopathy; improved formulations.
- Risks: Patent expirations; intense price competition from generics; shifts in treatment guidelines favoring other drug classes like ARNI (angiotensin receptor-neprilysin inhibitors).
Summary
Sales of COREG CR are projected to grow modestly over the next five years, driven by global cardiovascular disease prevalence, new formulation adoption, and guideline endorsements. Challenges include generic competition and patent expirations, which will influence pricing and market share trajectory.
Key Takeaways
- COREG CR faces significant cost competition due to generic carvedilol producers, limiting high-end pricing.
- Market expansion relies on new indications and approved markets, especially emerging economies.
- The drug’s sales growth remains moderate, averaging around 8-10% annually in coming years.
- Patent expirations diminish pricing power, necessitating product differentiation.
- Regulatory and guideline updates can materially impact sales dynamics.
FAQs
-
What factors influence COREG CR’s global market penetration?
Market penetration depends on regulatory approvals, physician prescribing habits, reimbursement policies, and patient adherence preferences.
-
How does generic competition affect COREG CR sales?
Generics offer lower-cost options, reducing the market share and revenue potential of branded COREG CR.
-
Are there upcoming regulations that could impact COREG CR sales?
Changes in cardiovascular treatment guidelines and approval of new indications can positively influence sales, while patent expirations and anti-price gouging measures can suppress revenue.
-
What regions offer the highest growth potential?
Emerging markets in Asia-Pacific, Latin America, and parts of Africa present opportunities due to expanding healthcare infrastructure and increasing cardiovascular disease prevalence.
-
How do treatment guidelines influence COREG CR sales?
Endorsements by AHA and ESC emphasize carvedilol’s role in certain heart failure and hypertension treatments, bolstering prescribing rates and sales.
Citations
[1] Medi-Tech Insights, 2022. "Global Cardiovascular Disease Market Overview."
[2] World Health Organization, 2021. "Global hypertension prevalence."
[3] IMS Health Reports, 2022. "Market trends for carvedilol and beta-blockers."
[4] American Heart Association, 2022. "Guidelines for the management of heart failure."
[5] U.S. Food and Drug Administration, 2019. "Market exclusivity periods for carvedilol formulations."