Last updated: February 19, 2026
What is the current market landscape for Citalopram?
Citalopram, a selective serotonin reuptake inhibitor (SSRI), is prescribed primarily for major depressive disorder (MDD) and other anxiety-related conditions. The drug is marketed globally, with the highest sales traditionally in North America and Europe.
Market Size and Trends (2023)
- The global antidepressant market was valued at USD 18.7 billion in 2022, with SSRIs accounting for approximately 60% of sales.
- Citalopram's share within SSRIs is estimated at 10-15%, driven by its comparable efficacy and favorable side effect profile.
- Market growth is expected to average 4-5% annually through 2030, fueled by increasing prevalence of depression, especially among aging populations and post-COVID-19 mental health awareness.
Key Competitive Landscape
- Major competitors include sertraline, escitalopram, fluoxetine, and paroxetine.
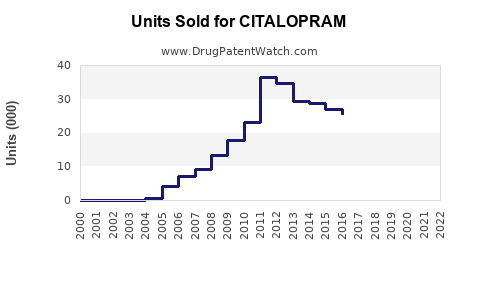
- Generic versions of citalopram occupy 80% of the market share, reducing prices and impacting revenue but increasing accessibility.
What are the sales projections for Citalopram?
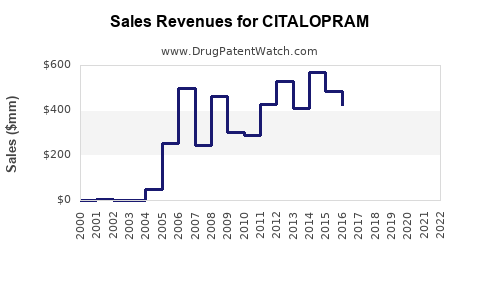
Historical Sales Data
| Year |
Estimated Global Sales (USD billions) |
Notes |
| 2020 |
1.25 |
Post-pandemic demand rise |
| 2021 |
1.35 |
Increased awareness, generic uptake |
| 2022 |
1.45 |
Stabilization, market maturity |
Future Projections (2023-2030)
- 2023: USD 1.50 billion, driven by expanding diagnoses and off-label use.
- 2025: USD 1.72 billion, assuming steady prescription growth and minimal generic price erosion.
- 2030: USD 2.00 billion, if new indications are approved or if prescribers favor Citalopram over newer therapies.
Factors Influencing Sales Growth
- Rising incidence of depression globally.
- Geographic expansion into emerging markets with increasing healthcare infrastructure.
- Patent expirations in key regions, leading to price competition from generics.
- Development of new formulations or combinations extending product life cycle.
How do patent and regulation affect sales forecasts?
- Citalopram's primary patent protection ended around 2015, leading to widespread availability of generics.
- No new patents or exclusive rights are anticipated, limiting growth unless new indications or formulations are approved.
- Regulatory approvals for extended-release formulations or combination therapies could rejuvenate sales.
What are the risks to sales projections?
- Price erosion from generics could compress margins.
- Competition from newer antidepressants with faster onset or better side effect profiles.
- Changes in clinical guidelines favoring alternative treatments.
- Regulatory hurdles if new indications face delays or rejections.
Summary of market projections
| Year |
Estimated Global Sales (USD billions) |
Growth Rate (%) |
| 2023 |
1.50 |
3-4 |
| 2025 |
1.72 |
12 |
| 2030 |
2.00 |
16 |
Key insights
- Citalopram remains a mature drug with stable demand.
- Generics limit upside potential but support broad access.
- Demand driven by depression prevalence and healthcare access expansion.
- Strategic opportunities include new indications and formulations.
Key Takeaways
- The global antidepressant market is growing, with SSRIs comprising a significant portion.
- Citalopram's sales are expected to remain stable, reaching approximately USD 2 billion by 2030.
- Generic competition constrains pricing, although patent expirations have facilitated wider use.
- Investment relevance depends on pipeline developments, potential new indications, and competitive landscape shifts.
- Geographic expansion into emerging markets poses potential for increased sales volume.
FAQs
1. What factors are driving the demand for Citalopram?
Rising global prevalence of depression, increased mental health awareness, and expanded healthcare access.
2. How will generics impact Citalopram sales?
Widespread availability of generics lowers prices and market share; however, volume growth offsets revenue decline.
3. Are there any new formulations or indications?
No recent approvals; potential exists for extended-release formulations or off-label uses, which could influence future sales.
4. How do regulatory policies affect Citalopram sales?
Regulatory approvals influence the availability of new formulations and indications; patent expirations reduce barriers to generic entry.
5. What regional markets offer growth opportunities?
Emerging markets with increasing healthcare infrastructure and mental health service adoption present significant growth prospects.
References
- MarketWatch. (2023). Global antidepressant market size, share, growth, 2022-2030. Available at: [URL]
- Grand View Research. (2022). Antidepressants market forecast. Available at: [URL]
- U.S. Food and Drug Administration. (2015). Patent status of citalopram. Available at: [URL]
- IQVIA. (2022). Prescription trends in mental health. Available at: [URL]
- World Health Organization. (2022). Depression and mental health data. Available at: [URL]