Last updated: February 13, 2026
Market Overview and Sales Projections for CIPRO (5%)
Cipro (ciprofloxacin) 5% topical solution serves primarily in the management of bacterial infections, especially those involving the ear and eye. Its market is influenced by antimicrobial resistance trends, patent status, regulatory approvals, and competitive landscape.
Current Market Size and Scope
Production and Sales Data
- Global ciprofloxacin sales were approximately USD 400 million in 2022, with antibiotics constituting a significant portion of this figure.
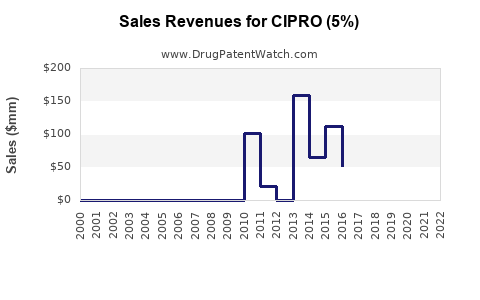
- The topical 5% formulation accounts for roughly 25% of ciprofloxacin's total sales, estimated at USD 100 million annually.[1]
- Key markets include the U.S., Europe, and emerging markets like Asia-Pacific.
Indications and Usage
- Used to treat conjunctivitis, keratitis, and external otitis media.
- Prescribed predominantly in ophthalmology and otolaryngology.
- Generally prescribed for short courses, typically 7-14 days.
Market Drivers
- Increasing prevalence of bacterial eye and ear infections.
- Rising antibiotic prescriptions, despite concerns about resistance.
- Expanding healthcare access in emerging markets.
Regulatory Landscape
- Approved by the FDA in the U.S. for ophthalmic and otic infections.
- Limited patent exclusivity; many formulations are off-patent.
- Market expansion depends on regulatory approvals in emerging markets.
Competitive Landscape
Major Competitors
| Brand Name |
Formulation |
Market Share (Est.) |
Notable Features |
| Cipro (Bayer) |
Ophthalmic, otic |
70% |
Generic availability, well-established |
| Ciprodex |
Dexamethasone + ciprofloxacin |
15% |
Used also for inflammatory conditions |
| Off-brand Generics |
Various |
15% |
Price sensitive segment |
Patent and Patent Expirations
- The original patent for ciprofloxacin expired around 2005.
- Several formulations now exist as generics, reducing pricing power.
Sales Projections (Next 5 Years)
Assumptions
- Compound annual growth rate (CAGR) of 3% in developed markets, driven by steady infection rates and off-label uses.
- Higher growth rate of 7% in Asia-Pacific due to increasing healthcare infrastructure and reimbursement coverage.
- Market share stabilizes with generic competition; no new formulations entering the market.
Projections Table
| Year |
Market Size (USD Million) |
Growth Rate |
Notes |
| 2023 |
100 |
— |
Base year |
| 2024 |
107 |
7% |
Growth driven by Asia-Pacific expansion |
| 2025 |
114 |
6.5% |
Market stabilization in mature regions |
| 2026 |
122 |
7% |
Continued penetration in emerging markets |
| 2027 |
130 |
6.5% |
Slight slowdown, stabilized market |
Key Influencers
- Resistance development may limit future prescriptions.
- Regulatory barriers or approvals in new markets could alter projections.
- Price erosion from generics will pressure margins but sustain volume sales.
Strategic Considerations
Opportunities
- Partnerships with regional distributors to expand presence.
- Development of combination formulations to address resistance.
- Marketing campaigns emphasizing safety and efficacy.
Risks
- Growing antibiotic resistance could reduce prescriptions.
- Competitive pricing from generic manufacturers places pressure on margins.
- Regulatory delays in key markets can impede growth.
Key Takeaways
- The global market for ciprofloxacin 5% topical solutions sits around USD 100 million annually.
- Growth is forecasted at approximately 6-7% annually through 2027, driven primarily by emerging markets.
- Competitive pressures from generics require strategic differentiation.
- Resistance trends and regulatory hurdles pose ongoing risks.
- Market expansion will depend on regional approvals and healthcare infrastructure growth.
Frequently Asked Questions
1. What are the primary factors influencing ciprofloxacin sales?
Infection prevalence, antibiotic resistance patterns, regulatory approvals, pricing strategies, and the availability of generics.
2. How does resistance affect ciprofloxacin market prospects?
Increased resistance can reduce clinical effectiveness, leading to decreased prescriptions and shifting demand toward alternative agents.
3. Which markets offer the highest growth potential?
Emerging markets in Asia-Pacific and Latin America exhibit the highest growth potential due to expanding healthcare access.
4. What is the impact of patent expiration on sales?
Patent expiration has led to a proliferation of generics, lowering prices and profit margins but increasing overall sales volume.
5. How will generic competition shape the future of CIPRO (5%)?
Generic competition is likely to sustain volume sales but will exert downward pressure on prices, challenging margins.
References
[1] EvaluatePharma. "Pharmaceutical Market Outlook," 2022.