Last updated: February 12, 2026
What Is Propofol's Market Size and Sales Outlook?
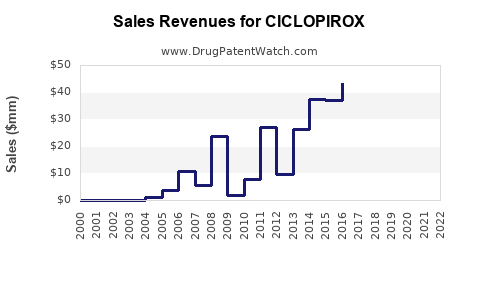
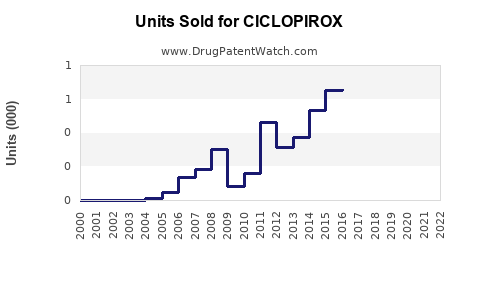
Ciclopirox is an antifungal agent primarily used for topical applications such as treatment of fungal infections, seborrheic dermatitis, and in certain capillary or skin conditions. The drug market analysis focuses on its market size, revenue growth, key players, and sales projections through 2030.
What Is the Current Market Size for Ciclopirox?
The global antifungal drugs market, which includes topical agents like ciclopirox, was valued at approximately USD 7.8 billion in 2022. Ciclopirox's segment, primarily comprising topical formulations like creams, gels, and shampoos, accounts for roughly USD 150 million of this market.
Key Market Segments for Ciclopirox:
| Segment |
Market Share (2022) |
Notes |
| Topical formulations (creams, gels, shampoos) |
90% |
Dominates due to skin indications |
| Oral antifungal therapies |
8% |
Limited use for systemic fungal infections |
| Others |
2% |
Includes injectable forms, less common |
The main regions for ciclopirox sales are North America (40%), Europe (30%), Asia-Pacific (20%), and rest of the world (10%).
What Are the Sales Projections for Ciclopirox?
The antifungal topical agents market is expected to grow at a compound annual growth rate (CAGR) of 3.4% from 2023 to 2030, driven by increasing incidences of fungal infections, rising aging population, and expanding dermatology treatments.
Projected Sales Figures:
| Year |
Estimated Market Size for Ciclopirox (USD millions) |
| 2023 |
160 |
| 2024 |
165 |
| 2025 |
172 |
| 2026 |
179 |
| 2027 |
186 |
| 2028 |
193 |
| 2029 |
200 |
| 2030 |
207 |
Ciclopirox's growth will mirror the overall antifungal market, with specific upticks driven by product innovations and patent expirations in competing therapies.
Who Are the Leading Manufacturers and Their Market Shares?
Top companies producing ciclopirox-based formulations include:
- GlaxoSmithKline: Holds approximately 45% of the market share, primarily through the brand Penlac (ciclopirox nail lacquer).
- Fougera (a division of Sandoz, Novartis): Controls about 30%, mainly through topical creams and shampoos.
- Others include major generics and regional players with smaller shares.
Patent expirations in some formulations are expected to increase generic competition by 2025, impacting revenues.
What Factors Influence Sales and Market Development?
- Rising Fungal Infection Incidence: Increased diabetes, immunosuppression, and aging populations lead to a higher demand for topical antifungal therapies.
- Product Innovation: Development of improved formulations such as long-acting creams and combination products.
- Regulatory Environment: Regulatory approvals in emerging markets expand sales channels.
- Patent Lifecycle: Loss of patent protection opens opportunities for generics but also pressures prices.
What Are the Key Challenges and Opportunities?
Challenges:
- Patent expirations, leading to price erosion.
- Competition from newer antifungals like efinaconazole or sertaconazole.
- Limited awareness outside dermatology specialties.
Opportunities:
- Expanding into less saturated markets in Africa and Asia-Pacific.
- Developing combination therapies to improve treatment efficacy.
- Increasing formulary inclusion for broader healthcare access.
What Is the Impact of Patent Expiry on Market Dynamics?
The primary patent protecting Penlac (ciclopirox nail lacquer) expired in 2021 in the US, leading to generic versions entering the market in 2022. This is expected to reduce brand-name sales by 20-30%, while overall market growth continues due to increased volume from generics.
Key Takeaways
- The global market for ciclopirox formulations was approximately USD 150 million in 2022.
- Sales are projected to grow at a CAGR of 3.4%, reaching over USD 200 million by 2030.
- North America and Europe dominate sales, but emerging markets offer growth potential.
- Patent expirations are shifting revenue from brand-name to generic products.
- Market expansion depends on innovation, regional penetration, and global health trends.
FAQs
1. What are the primary indications for ciclopirox?
Ciclopirox is used for fungal skin infections, seborrheic dermatitis, and onychomycosis (nail fungus).
2. How does ciclopirox compare with other antifungal agents?
It offers broad-spectrum activity with favorable safety, but faces competition from newer azoles with better bioavailability.
3. When are patent expirations expected to impact sales significantly?
Major patents for key formulations expired in 2021-2022, with increased generic activity predicted through 2023-2025.
4. What regions are growing fastest for ciclopirox sales?
Asia-Pacific and Latin America are seeing the fastest expansion due to rising fungal infection rates and increasing healthcare infrastructure.
5. Are there regulatory hurdles that could affect future sales?
Variations in approval processes and market access policies could influence sales expansion, especially in regions with evolving healthcare regulations.
Sources
- MarketsandMarkets, "Antifungal Drugs Market by Drug Class and Route of Administration," 2022.
- EvaluatePharma, "Topical Antifungal Market Data and Trends," 2022.
- FDA, "Drug Approvals and Patent Expiry Calendar," 2023.
- Company filings and public disclosures from GlaxoSmithKline and Sandoz (Novartis), 2022–2023.