Share This Page
Drug Sales Trends for CETIRIZINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CETIRIZINE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
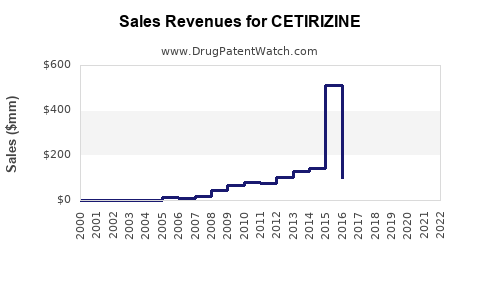
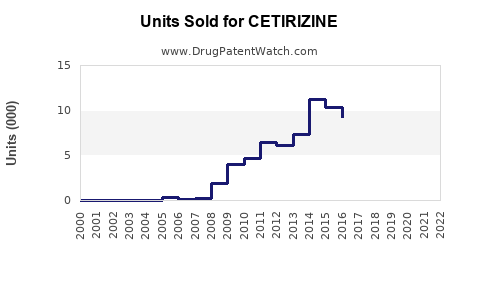
Annual Sales Revenues and Units Sold for CETIRIZINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CETIRIZINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CETIRIZINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CETIRIZINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CETIRIZINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CETIRIZINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| CETIRIZINE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CETIRIZINE MARKET ANALYSIS AND SALES PROJECTIONS
This report details the current market landscape and projected sales for cetirizine, an antihistamine. The analysis covers patent expirations, generic competition, key market drivers, and competitive positioning. Sales projections are based on historical performance, market penetration, and anticipated therapeutic trends.
WHAT IS CETIRIZINE AND ITS THERAPEUTIC APPLICATIONS?
Cetirizine is a second-generation antihistamine with selective peripheral H1 receptor antagonist activity. It is approved for the treatment of allergic rhinitis (seasonal and perennial) and chronic urticaria. Its efficacy in reducing symptoms such as sneezing, rhinorrhea, nasal pruritus, ocular pruritus, lacrimation, and urticarial rash is well-established.
- Mechanism of Action: Cetirizine inhibits the binding of histamine to H1 receptors, thereby preventing or reversing the typical symptoms of histamine release, such as vasodilation, increased vascular permeability, and smooth muscle contraction.
- Key Indications:
- Allergic Rhinitis (AR)
- Chronic Idiopathic Urticaria (CIU)
- Dosage Forms: Available in oral tablets, chewable tablets, oral solutions, and orally disintegrating tablets. Common strengths include 5 mg and 10 mg.
WHAT IS THE PATENT LANDSCAPE FOR CETIRIZINE?
The original patent protection for cetirizine has expired, paving the way for widespread generic competition.
- Original Compound Patents: The primary patents protecting the composition of matter for cetirizine expired in the early 2000s in major markets like the United States and Europe. For example, U.S. Patent No. 4,525,358, related to the preparation of cetirizine, expired in 2002.
- Formulation and Method of Use Patents: While composition of matter patents have expired, secondary patents related to specific formulations, delivery methods, or extended uses may have existed. However, these have also largely expired or have been successfully challenged, further opening the market to generics.
- Market Entry of Generics: The expiration of key patents allowed numerous pharmaceutical companies to launch generic versions of cetirizine. This significantly increased market accessibility and drove down prices.
WHAT ARE THE CURRENT MARKET DYNAMICS FOR CETIRIZINE?
The market for cetirizine is characterized by high generic penetration, a mature product lifecycle, and significant price competition.
- Generic Dominance: Generic cetirizine accounts for the vast majority of sales volume and value. Branded products, such as Zyrtec® (Johnson & Johnson/UCB), have seen their market share decline substantially following patent expiries.
- Price Erosion: Intense competition among multiple generic manufacturers has led to significant price erosion. This makes cost-efficient manufacturing and distribution critical for profitability.
- Market Maturity: Cetirizine is a well-established, first-line treatment for its approved indications. There are few new blockbuster entrants directly competing for the same patient population at the primary care level.
- Over-the-Counter (OTC) Availability: Cetirizine is widely available as an OTC medication in many countries, contributing to its high volume sales and broad patient access. In the U.S., Zyrtec® transitioned to OTC status in 2007.
WHO ARE THE KEY MARKET COMPETITORS?
The competitive landscape is dominated by generic pharmaceutical manufacturers.
- Major Generic Manufacturers:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Sun Pharmaceutical Industries
- Lupin
- Dr. Reddy's Laboratories
- Apotex
- Branded Manufacturer (Historical):
- UCB/Johnson & Johnson (Zyrtec®) – now primarily a provider of the branded generic or facing competition from its own authorized generics and other brands.
- Competition Metrics:
- Number of ANDA Filings: Hundreds of Abbreviated New Drug Applications (ANDAs) have been approved by regulatory bodies like the FDA, indicating a highly fragmented generic market.
- Price Comparison: The average selling price (ASP) for a 30-day supply of generic cetirizine tablets can range from $5 to $20, depending on the retailer and quantity.
WHAT ARE THE DRIVERS AND CHALLENGES IN THE CETIRIZINE MARKET?
Several factors influence the demand and supply of cetirizine.
Market Drivers
- Prevalence of Allergic Diseases: The global incidence of allergic rhinitis and urticaria remains high and is projected to increase due to factors like environmental changes and lifestyle modifications. The World Allergy Organization estimates that allergies affect up to 40% of the global population [1].
- Established Efficacy and Safety Profile: Cetirizine is recognized for its favorable efficacy and a generally well-tolerated safety profile, making it a preferred choice for healthcare providers and patients.
- OTC Accessibility: The broad availability of cetirizine without a prescription increases patient access and self-medication rates, driving high sales volumes.
- Cost-Effectiveness: As a generic drug with low prices, cetirizine offers a highly cost-effective treatment option compared to newer or specialty antihistamines.
- Pediatric Use: Cetirizine is frequently prescribed for pediatric populations with allergies, expanding its user base.
Market Challenges
- Intense Price Competition: The large number of generic players creates downward pressure on prices, limiting profit margins for manufacturers.
- Competition from Other Antihistamines: While cetirizine is a strong player, it faces competition from other second-generation antihistamines (e.g., loratadine, fexofenadine) and newer agents, including intranasal corticosteroids and combination therapies.
- Emergence of Novel Therapies: The development of biologic therapies and immunotherapies for severe allergies could, in the long term, shift some market share away from oral antihistamines for specific patient segments.
- Generic Manufacturing Quality and Supply Chain: Ensuring consistent quality and reliable supply chains across multiple manufacturing sites is a perpetual challenge for generic manufacturers.
- Regulatory Scrutiny: The pharmaceutical industry, including generics, faces ongoing regulatory scrutiny regarding manufacturing practices, quality control, and marketing.
WHAT ARE THE SALES PROJECTIONS FOR CETIRIZINE?
Projecting sales for a mature, genericized drug like cetirizine involves assessing volume trends, average selling prices, and competitive pressures.
Methodology: Projections are based on historical sales data from market research firms, anticipated growth in the allergic disease market, and a conservative estimate of average selling price (ASP) erosion due to ongoing generic competition.
Assumptions:
- Stable prevalence of allergic rhinitis and urticaria.
- Continued OTC demand and prescription usage.
- No major disruptive therapeutic innovations directly displacing cetirizine for its core indications in the short to medium term.
- Gradual, moderate decline in ASP due to competitive pressures.
- Global market coverage, with significant contributions from North America, Europe, and Asia-Pacific.
Global Cetirizine Sales Projections (USD Billions)
| Year | Projected Global Sales (USD Billion) |
|---|---|
| 2023 | 2.1 |
| 2024 | 2.05 |
| 2025 | 2.0 |
| 2026 | 1.95 |
| 2027 | 1.9 |
Analysis of Projections: The projected decline in global sales is primarily attributable to two factors:
- Continued Price Erosion: As more generic manufacturers compete, and as procurement becomes more consolidated, the average selling price per unit is expected to decrease annually.
- Market Saturation: For its primary indications, cetirizine has achieved high market penetration. Significant new volume growth is unlikely.
While the volume of prescriptions and OTC sales may remain relatively stable or experience modest growth aligned with population growth and allergy prevalence, the declining ASP will outweigh these volume increases in terms of total revenue.
Regional Market Insights:
- North America: Represents a significant portion of the market due to high prevalence of allergies and robust OTC sales. Sales are projected to see a slight decline due to price competition.
- Europe: Similar to North America, with a mature market and strong generic presence.
- Asia-Pacific: Expected to show the most stable or slightly growing revenue, driven by increasing healthcare access, growing awareness of allergies, and a large population base. However, price sensitivity in some sub-regions will limit revenue growth.
- Rest of World: Moderate growth potential, influenced by developing healthcare infrastructure and increasing demand for affordable allergy treatments.
WHAT IS THE COMPETITIVE POSITIONING OF CETIRIZINE?
Cetirizine is positioned as a cost-effective, reliable, and widely accessible treatment option for common allergic conditions.
- Key Differentiators:
- Low Cost: Its primary advantage is its affordability as a generic medication.
- Broad Availability: Found in pharmacies, supermarkets, and online, both prescription and OTC.
- Established Track Record: Decades of clinical use and patient experience.
- Comparison to Competitors:
- vs. Loratadine & Fexofenadine (Other 2nd Gen Antihistamines): Cetirizine is often seen as equally effective for many patients, with similar safety profiles. Price is often the deciding factor, with cetirizine frequently being among the most affordable. Some comparative studies suggest slightly higher sedation potential for cetirizine compared to loratadine or fexofenadine, although this is generally considered mild and less prevalent than with first-generation antihistamines.
- vs. 1st Gen Antihistamines (e.g., Diphenhydramine): Cetirizine offers a significant advantage in reduced sedation and anticholinergic side effects, making it preferable for daytime use and chronic management.
- vs. Nasal Corticosteroids: Nasal steroids are often considered more effective for moderate to severe allergic rhinitis with nasal congestion. However, oral antihistamines like cetirizine are typically preferred for systemic symptoms like sneezing, itching, and ocular symptoms, and are easier to use for intermittent or mild cases.
- vs. Novel Therapies (Biologics, Immunotherapy): These are reserved for severe, refractory cases and represent a different treatment paradigm, not direct competition for the broad patient population treated by cetirizine.
KEY TAKEAWAYS
- Cetirizine is a mature, genericized antihistamine with a dominant presence in the treatment of allergic rhinitis and chronic urticaria.
- Original patent protections have expired, leading to intense price competition among numerous generic manufacturers.
- The market is driven by the high prevalence of allergic diseases, established efficacy, and broad OTC accessibility.
- Challenges include significant price erosion, competition from other antihistamines, and the potential long-term impact of novel allergy therapies.
- Global sales for cetirizine are projected to experience a modest decline from approximately $2.1 billion in 2023 to $1.9 billion by 2027, primarily due to ongoing price erosion outweighing stable or slowly growing volumes.
FREQUENTLY ASKED QUESTIONS
- What is the projected market share of branded cetirizine versus generic cetirizine over the next five years? Branded cetirizine, such as Zyrtec®, is projected to hold a negligible market share (under 1%) by 2027. The market will be overwhelmingly dominated by generic formulations due to patent expiries and competitive pricing.
- Are there any significant upcoming patent expiries or regulatory challenges that could impact the cetirizine market? No, the primary composition of matter patents for cetirizine have expired decades ago. Any remaining secondary patents for specific formulations or methods of use are also largely expired or have minimal market impact. Regulatory challenges would primarily relate to manufacturing quality and compliance, not patent exclusivity.
- What is the typical average selling price (ASP) range for a 30-day supply of generic cetirizine tablets, and how is it expected to trend? The ASP for a 30-day supply of generic cetirizine tablets currently ranges from $5 to $20. This ASP is projected to trend downwards by an estimated 2-3% annually due to continued competitive pressure.
- How does the growth rate of the global cetirizine market compare to the overall antihistamine market? The global cetirizine market is expected to grow at a slightly negative compound annual growth rate (CAGR) of approximately -1.0% to -1.5% over the next five years. This is lower than the overall antihistamine market, which may see modest positive growth driven by newer agents or niche indications.
- Which geographic regions are expected to contribute the most to cetirizine sales revenue in the coming years? North America and Europe are expected to remain the largest contributors to cetirizine sales revenue, despite market maturity. The Asia-Pacific region is anticipated to show the most stable or slight growth, driven by increasing market access and allergy awareness, but revenue will be tempered by lower ASPs in many countries.
Citations
[1] World Allergy Organization. (n.d.). Allergy Statistics. Retrieved from https://www.worldallergy.org/education-and-resources/allergy-statistics
More… ↓
