Last updated: July 27, 2025
Here is a comprehensive market analysis and sales projections for Catapres-TTS (clonidine transdermal patch):
Market Overview
Catapres-TTS is a transdermal patch formulation of clonidine, primarily used to treat hypertension. Key points about the market:
-
The global transdermal patch market is projected to reach over $15 billion by 2026, growing at a CAGR of around 4-5%[1].
-
Clonidine patches like Catapres-TTS occupy a niche segment within this broader market, focused on treating hypertension and some off-label uses.
-
The addressable market for Catapres-TTS is estimated at around $930 million in Europe, specifically for resistant hypertension patients[3].
-
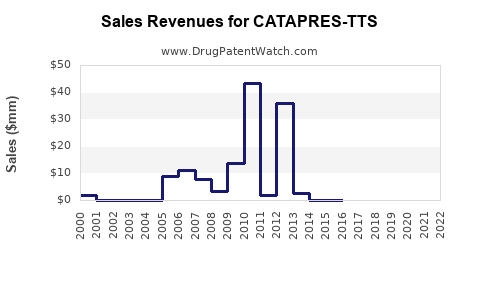
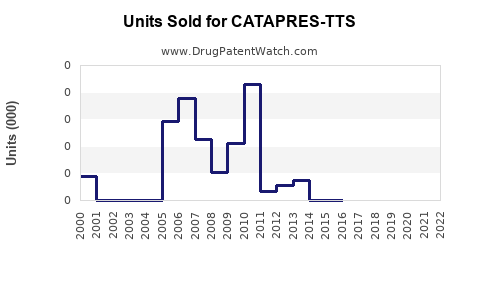
In the U.S., Catapres-TTS had total sales of approximately $180 million in 2014 before generic entry[2][5].
Sales Projections
Based on the available data, here are sales projections for Catapres-TTS:
-
Global sales are likely declining due to generic competition, estimated at $50-100 million annually.
-
In Europe, there is potential to grow sales to €100-200 million by capturing more of the €930 million addressable market[3].
-
U.S. brand sales have likely declined substantially from the $180 million peak in 2014 due to generics.
-
Emerging markets present growth opportunities as hypertension prevalence rises.
Key Market Dynamics
Several factors are shaping the market for Catapres-TTS:
-
Generic competition: Multiple generic versions have entered the market since 2014, putting pressure on brand sales and pricing[2][5].
-
Aging population: An older demographic is increasing the prevalence of hypertension globally.
-
Advantages of patch formulation: Once-weekly dosing improves patient compliance compared to oral medications.
-
Expanding indications: Off-label use for conditions like ADHD presents growth opportunities[6].
-
Pricing pressures: Healthcare cost containment efforts are limiting price growth potential.
Competitive Landscape
The market for clonidine patches is moderately competitive:
-
Generic manufacturers like Actavis (now Teva) have captured significant market share[2][5].
-
Other antihypertensive medications provide indirect competition.
-
Innovative drug delivery systems could disrupt the market.
Growth Opportunities
Potential areas for growth include:
-
Geographic expansion, especially in emerging markets with rising hypertension rates.
-
New indications like ADHD, menopausal symptoms, and pain management[6].
-
Improved formulations with better side effect profiles or longer duration of action.
-
Combination therapies with other antihypertensive agents.
Challenges
Key challenges facing Catapres-TTS include:
-
Continued erosion from generic competition.
-
Side effects like skin reactions limiting long-term use[7].
-
Regulatory hurdles for label expansions or new formulations.
-
Physician preference for newer antihypertensive drug classes.
In conclusion, while Catapres-TTS faces headwinds from generic competition, opportunities exist to stabilize and potentially grow sales through geographic expansion, new indications, and formulation improvements. Focusing on its unique advantages in patient compliance could help differentiate it in the crowded antihypertensive market.