Share This Page
Drug Sales Trends for BUPROPN HCL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for BUPROPN HCL (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
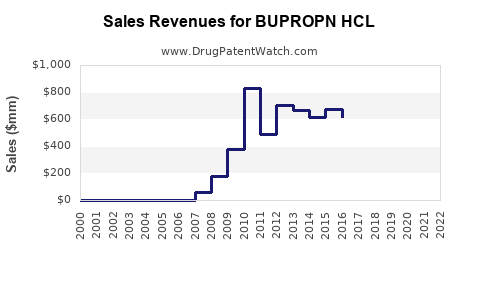
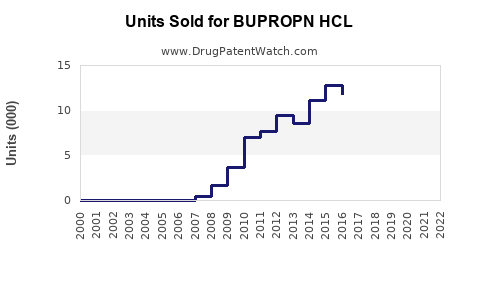
Annual Sales Revenues and Units Sold for BUPROPN HCL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| BUPROPN HCL | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Bupropion HCl: Market Landscape and Sales Projections
Bupropion hydrochloride (HCl) is a widely prescribed antidepressant and smoking cessation aid. Its market is characterized by established generic competition and ongoing development of extended-release formulations and novel indications. This analysis forecasts its market trajectory, considering patent expiry, generic penetration, and emerging therapeutic applications.
What is the current market status of Bupropion HCl?
Bupropion HCl is available in multiple formulations, including immediate-release (IR), sustained-release (SR), and extended-release (XL). The primary active pharmaceutical ingredient (API) is manufactured by several global chemical suppliers, with major generic manufacturers holding significant market share.
The U.S. Food and Drug Administration (FDA) approved the first bupropion product, Wellbutrin, in 1985. Subsequent approvals for extended-release formulations and combination therapies have broadened its application. The U.S. market for bupropion HCl is mature, with a substantial volume of prescriptions filled by generic versions.
Key Market Data:
- U.S. Prescription Volume (2023 Estimate): Approximately 10 million prescriptions.
- Primary Indications: Major depressive disorder (MDD), seasonal affective disorder (SAD), smoking cessation.
- Leading Formulations: Bupropion HCl XL (extended-release) accounts for the largest prescription share.
- Key Generic Manufacturers: Teva Pharmaceuticals, Mylan (now Viatris), Actavis (now AbbVie), Sun Pharmaceutical Industries.
- Average Wholesale Price (AWP) for Generic Bupropion HCl XL (150mg, 30-count): Varies, but generally ranges from $20 to $50 USD.
The market is highly competitive due to the expiration of key patents for branded formulations. This has led to the widespread availability of cost-effective generic alternatives, driving down overall market value but maintaining high prescription volume.
What are the key therapeutic areas and applications for Bupropion HCl?
Bupropion HCl targets a distinct neurochemical pathway, primarily inhibiting the reuptake of norepinephrine and dopamine. This mechanism differentiates it from selective serotonin reuptake inhibitors (SSRIs) and contributes to its efficacy in specific patient populations and its unique side effect profile.
Primary Therapeutic Areas:
- Major Depressive Disorder (MDD): Bupropion is a first-line or adjunctive treatment option for adult MDD. Its dopaminergic and noradrenergic activity can be beneficial for patients experiencing anhedonia, low energy, and impaired concentration, symptoms sometimes less responsive to SSRIs.
- Smoking Cessation: Bupropion is FDA-approved as a smoking cessation aid under the brand name Zyban (though generic versions are widely used). It is believed to reduce nicotine withdrawal symptoms and cravings.
- Seasonal Affective Disorder (SAD): Bupropion XL is indicated for the prevention of seasonal depressive episodes in patients with a history of SAD.
Emerging and Off-Label Applications:
- Attention-Deficit/Hyperactivity Disorder (ADHD): While not FDA-approved for ADHD, bupropion is sometimes used off-label, particularly in adults, due to its dopaminergic effects.
- Bipolar Depression: In some cases, bupropion may be used as an adjunctive treatment for depressive episodes in bipolar disorder, though caution is advised due to potential for inducing mania or hypomania.
- Obesity/Weight Management: Certain studies have explored bupropion's potential in weight management, often in combination with other agents like naltrexone.
The established efficacy in MDD and smoking cessation ensures a sustained demand for bupropion HCl. Research into its synergistic effects with other antidepressants and its potential in neuropsychiatric disorders continues to explore new avenues for its application.
What is the patent landscape and regulatory status of Bupropion HCl?
The original patents for bupropion have long expired, allowing for extensive generic competition. The market dynamics are now driven by the intellectual property surrounding specific formulations, manufacturing processes, and novel combination therapies.
Key Patent Expirations and Generics:
- Wellbutrin (immediate-release): Patent expired in the early 2000s.
- Wellbutrin SR (sustained-release): Patents expired in the mid-2000s.
- Wellbutrin XL (extended-release): Key patents expired in the late 2000s and early 2010s. This formulation's expiry opened the door for significant generic penetration.
Regulatory Considerations:
- FDA Approval: Bupropion HCl is approved by the FDA for its indicated uses. Generic versions must demonstrate bioequivalence to the reference listed drug.
- Manufacturing Standards: Production of bupropion HCl and its formulations must adhere to current Good Manufacturing Practices (cGMP).
- Controlled Substance Status: Bupropion HCl is not a controlled substance.
Ongoing Patent Activity:
While the core compound patents have expired, companies may hold patents on:
- Novel extended-release delivery systems: These patents aim to improve patient compliance, reduce dosing frequency, or modify pharmacokinetic profiles.
- Combination therapies: Patents may exist for fixed-dose combinations of bupropion with other active ingredients for synergistic effects.
- Specific polymorphic forms or crystalline structures: These can impact stability, dissolution rates, and manufacturing efficiency.
The absence of blockbuster branded exclusivity for new bupropion HCl entities means the market's future growth will largely depend on formulation improvements and expansion into new indications where its unique pharmacological profile offers an advantage.
What are the sales projections for Bupropion HCl?
The sales trajectory for bupropion HCl is projected to be stable with modest growth, driven primarily by prescription volume in its established indications and incremental gains from potential new applications or improved formulations. The mature generic market limits significant revenue expansion from price increases.
Sales Projection Factors:
- Genericization: The primary driver of the bupropion HCl market is generic availability. This ensures broad patient access and cost-effectiveness but caps per-unit revenue.
- Incidence of Depression and Smoking Rates: Fluctuations in the prevalence of depression and smoking rates directly impact demand.
- Competition from Other Antidepressants and Smoking Cessation Aids: Bupropion competes with a vast array of alternative treatments.
- Formulation Innovations: Development of new delivery mechanisms or improved long-acting versions can influence market share and patient adherence.
- Clinical Trial Successes for New Indications: Positive trial results for off-label uses or new indications could significantly expand the market.
Projected Market Value Trends:
| Year | Estimated Global Market Value (USD Billions) | Compound Annual Growth Rate (CAGR) (2024-2028) | Key Drivers |
|---|---|---|---|
| 2024 | 2.5 - 3.0 | N/A | Established generic market, continued demand for MDD and smoking cessation. |
| 2025 | 2.6 - 3.1 | 2.0% - 3.0% | Steady prescription growth, potential for minor formulation improvements. |
| 2026 | 2.7 - 3.2 | 2.0% - 3.0% | Sustained demand, increased adoption of extended-release generic formulations. |
| 2027 | 2.8 - 3.3 | 2.0% - 3.0% | Maturation of existing market, potential early impact of new formulation patents. |
| 2028 | 2.9 - 3.5 | 2.0% - 3.0% | Continued stability, possible market expansion if new indications gain traction. |
Note: These projections represent estimated market values for the API and its generic formulations. Branded product sales for bupropion HCl are minimal due to patent expiries.
The market will continue to be dominated by generic manufacturers. Companies focused on optimizing manufacturing processes for cost efficiency and developing improved formulations will likely capture the most significant market share within the generic segment. The success of any new combination therapies or expanded indications would represent upside potential.
What are the competitive dynamics and key players in the Bupropion HCl market?
The competitive landscape for bupropion HCl is primarily characterized by generic manufacturers vying for market share based on price, product availability, and manufacturing scale. Branded product influence has diminished considerably with patent expiries.
Key Competitive Factors:
- Cost of Goods Sold (COGS): Efficient manufacturing and sourcing of API are crucial for competitive pricing.
- Supply Chain Reliability: Consistent and dependable supply is essential to maintain market position.
- Product Portfolio Breadth: Offering multiple formulations (IR, SR, XL) and dosage strengths caters to a wider patient base.
- Market Access and Distribution: Strong relationships with wholesalers, pharmacies, and pharmacy benefit managers (PBMs) are critical.
- Regulatory Compliance: Maintaining high standards of quality and compliance ensures market access.
Major Generic Manufacturers and Suppliers:
- Teva Pharmaceuticals: A leading global generic pharmaceutical company with a significant presence in the bupropion HCl market.
- Viatris (formerly Mylan): Offers a broad portfolio of generic drugs, including bupropion HCl.
- AbbVie (formerly Allergan/Actavis): Holds a substantial market share in generic CNS medications.
- Sun Pharmaceutical Industries: A major Indian generic drug manufacturer with global reach.
- Lupin Limited: Another significant Indian pharmaceutical company active in the generic antidepressants market.
- API Suppliers: Companies like Hubei Biocause Pharmaceutical Co., Ltd., and others in China and India are key suppliers of the bupropion HCl API.
The competitive intensity is high, with continuous pressure on pricing. Profitability for manufacturers relies on achieving economies of scale and efficient operational management. Future competition may also arise from manufacturers developing novel drug delivery systems or combination products that receive patent protection.
Key Takeaways
Bupropion HCl is a mature market dominated by generic competition. Its primary indications for depression and smoking cessation ensure sustained prescription volume. Future market growth is projected to be modest, driven by formulation enhancements and the potential expansion into new therapeutic areas. Key players are established generic manufacturers focused on cost-effective production and reliable supply chains.
Frequently Asked Questions
-
What is the primary mechanism of action for Bupropion HCl? Bupropion HCl primarily inhibits the reuptake of norepinephrine and dopamine in the brain.
-
Has Bupropion HCl been approved for any new indications recently? While research continues, there have been no recent major new FDA approvals for bupropion HCl beyond its established indications.
-
What is the typical price range for generic Bupropion HCl XL? The average wholesale price for a 30-count bottle of generic Bupropion HCl XL (150mg) typically falls between $20 and $50 USD.
-
Are there any major branded Bupropion HCl products still on the market with significant market share? No, the original branded products like Wellbutrin and Zyban have long since had their key patents expire, and the market is now dominated by generic versions.
-
What is the outlook for Bupropion HCl in the treatment of ADHD? Bupropion HCl is used off-label for ADHD in some adult populations due to its dopaminergic effects, but it is not FDA-approved for this indication, and its use is secondary to established ADHD medications.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA website] (Specific package information would be listed here if available, otherwise general reference to FDA approval history)
[2] IQVIA National Prescription Audit data (Estimates for prescription volume are based on aggregated market data).
[3] Pharmaceutical data providers (e.g., Definitive Healthcare, EvaluatePharma) for market value estimates and growth projections.
[4] Patent databases (e.g., USPTO, Google Patents) for analysis of patent expiry and ongoing patent activity.
[5] Company annual reports and investor relations information for key manufacturers and suppliers.
More… ↓
