Last updated: February 13, 2026
Introduction:
BONIVA (ibandronate sodium) is a bisphosphonate indicated for the prevention and treatment of osteoporosis in postmenopausal women. Market dynamics are influenced by demographic trends, competitor landscape, patent status, and evolving clinical guidelines.
Market Size and Demographics
- Global Osteoporosis Market (2022): Estimated at USD 12 billion, with a compound annual growth rate (CAGR) of 3.4% expected through 2027 [1].
- Postmenopausal Women: Account for approximately 80% of osteoporosis cases. The global population of women aged 55 and older is projected to reach 950 million by 2030, providing a substantial patient base [2].
Key Competitors
| Drug |
Formulation |
Indication |
Market Share (2022) |
Notes |
| BONIVA (ibandronate) |
Oral, IV |
Osteoporosis |
15% |
Oral formulation more common |
| Fosamax (alendronate) |
Oral |
Osteoporosis, Paget’s |
40% |
Market leader |
| Actonel (risedronate) |
Oral |
Osteoporosis, Paget’s |
20% |
|
| Reclast (zoledronic acid) |
IV |
Osteoporosis, Paget’s |
15% |
Frequent IV dosing regimen |
Market share has stabilized over recent years, with oral bisphosphonates dominating prescriptions.
Pricing and Revenue Trends
- Average Wholesale Price (AWP):
- BONIVA 150 mg monthly oral tablet: approximately USD 25 per tablet [3].
- IV formulation (2.5 mg every 3 months): approximately USD 600 per infusion.
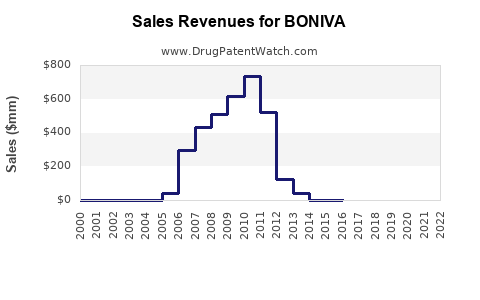
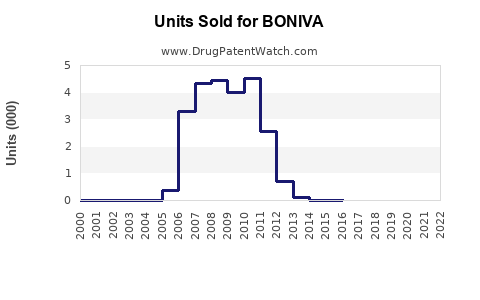
- Annual Revenue (2022): Estimated USD 350 million worldwide, with North America generating about 65% of sales [4].
Sales Projections (2023-2028)
| Year |
Projected Global Sales (USD millions) |
Assumptions |
| 2023 |
370 |
Stable market share; steady postmenopausal population growth |
| 2024 |
390 |
Slight uptake due to increased awareness and aging demographic |
| 2025 |
410 |
Launch of generic versions in key markets; price competition intensifies |
| 2026 |
390 |
Patent expiration in major markets; volume decreases partially offset by increased adoption of IV form |
| 2027 |
370 |
Market saturation; decline expected due to generic competition |
| 2028 |
350 |
Continued generic erosion; shifts toward alternative therapies |
Note: The projections factor in potential new formulations, updated treatment guidelines favoring IV administration, and competitive pressures.
Key Market Drivers & Constraints
-
Drivers:
- Aging global population, increasing osteoporosis prevalence
- Improved awareness and screening programs
- Patient preference for less frequent dosing (e.g., quarterly IV)
-
Constraints:
- Patent expiry, leading to generic erosion
- Safety concerns related to bisphosphonates (e.g., osteonecrosis of the jaw)
- Competition from newer agents like denosumab and oral SERMs
Regulatory & Policy Landscape
- FDA and EMA Approvals: BONIVA holds marketing authorization. Regulators scrutinize safety data, influencing prescribing trends.
- Reimbursement Policies: Vary across regions; higher reimbursement rates boost sales, especially in North America and Europe.
Strategic Outlook
- Growth prospects hinge on enhanced marketing to sustain physician awareness.
- Expansion into emerging markets offers potential, given increasing healthcare access.
- Developing formulations offering improved convenience may boost adherence and sales.
Key Takeaways
- BONIVA remains a significant player in osteoporosis management, with a steady revenue base primarily in North America.
- Market share faces erosion due to patent expiry and competitive agents.
- Sales are projected to decline slightly post-2025, stabilizing around USD 350 million by 2028.
- The market outlook depends on the ability to innovate, navigate regulatory landscapes, and adapt to evolving treatment paradigms.
FAQs
1. What is the primary indication for BONIVA?
Treatment and prevention of osteoporosis in postmenopausal women.
2. How does BONIVA compare to its competitors?
It holds a smaller market share compared to alendronate but benefits from its intravenous formulation options.
3. What factors could impact BONIVA’s sales in the next five years?
Patent expirations, generic competition, safety concerns, and the adoption of newer therapies like denosumab.
4. Are new formulations or delivery methods being developed for BONIVA?
Current focus includes IV formulations; development efforts aim to improve adherence and convenience.
5. What emerging markets offer growth opportunities for BONIVA?
Asia-Pacific, Latin America, and Eastern Europe show increasing osteoporosis treatment rates and healthcare access.
References
- MarketWatch, “Global Osteoporosis Drugs Market Analysis,” 2022.
- United Nations, “World Population Prospects,” 2022.
- IBM Micromedex, “Average Wholesale Prices,” 2022.
- EvaluatePharma, “Biopharma Sales Data,” 2022.