Share This Page
Drug Sales Trends for BETADINE
✉ Email this page to a colleague
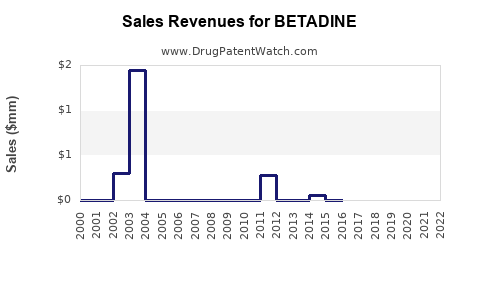
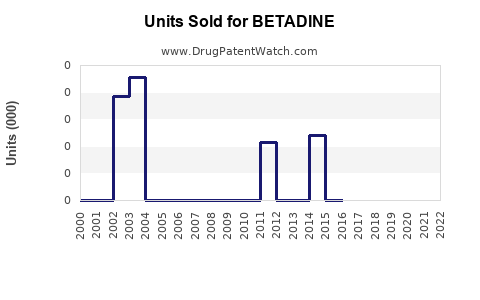
Annual Sales Revenues and Units Sold for BETADINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BETADINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BETADINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BETADINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Betadine Market Analysis and Sales Projections
Overview
Betadine, a topical antiseptic containing povidone-iodine, has maintained a significant presence in medical and consumer markets since its introduction. Its main applications involve skin disinfection, wound cleaning, and preoperative skin preparation. The global antiseptic market grows at roughly 4-6% annually, driven by expanding healthcare infrastructure, rising surgical volumes, and increasing awareness about infection control.
Market Size and Segmentation
Global Market Valuation
As of 2022, the global antiseptic market was valued at approximately USD 8 billion. Betadine is among the leading iodine-based antiseptics registering a domestic and international presence.
Segment Breakdown
The antiseptic segment, dominated by iodine compounds like Betadine, comprises:
| Segment | Share (%) | Description |
|---|---|---|
| Surgical preparation | 40% | Skin prep for surgeries and invasive procedures |
| Wound cleansing | 25% | First aid, minor cuts, and abrasions |
| Consumer antiseptics | 20% | Over-the-counter disinfection products |
| Others | 15% | Dental, gynecological, institutional uses |
Regional Distribution
The largest markets include North America (35%), Europe (20%), Asia-Pacific (30%), Latin America (8%), and Middle East & Africa (7%). Asia-Pacific exhibits the highest growth rate (~7%) attributable to expanding healthcare infrastructure and increasing surgical procedures.
Competitive Landscape
Major competitors include:
- Betadine (Mundipharma, Johnson & Johnson)
- Lucasol (Colgate-Palmolive)
- Isodine (S. C. Johnson)
- Babies' antiseptics brands
Betadine’s market position remains strong due to its brand recognition, wide product range (solutions, ointments, wipes), and established supply chain.
Regulatory and Policy Factors
- FDA approval (U.S.) for topical antiseptics including Betadine means off-label and new indications are restricted.
- EMA approvals in Europe align with same standards, facilitating cross-border sales.
- Asian Regulatory environments vary; India’s Drugs and Cosmetics Act permits OTC use of Betadine for minor cuts.
Trade and reimbursement policies influence market penetration, especially in emerging markets where healthcare expenditures are increasing.
Sales Trends and Projections
Historical Sales Data (2018-2022)
- North America: USD 1.5 billion (2022), with a CAGR of 2.5%
- Europe: USD 1.2 billion, CAGR of 2.0%
- Asia-Pacific: USD 2.4 billion, CAGR of 7.0%
- Rest of World: USD 1.1 billion, CAGR of 3.2%
Projected 2023-2028 Growth
Assuming steady growth, driven by increased healthcare activity and infection control awareness:
| Year | Estimated Market Value (USD billion) | Growth Rate (%) |
|---|---|---|
| 2023 | 9.0 | 6.2 |
| 2024 | 9.55 | 6.1 |
| 2025 | 10.12 | 6.0 |
| 2026 | 10.72 | 5.9 |
| 2027 | 11.36 | 5.9 |
| 2028 | 12.03 | 6.0 |
Key Drivers for Sales Growth
- Increasing surgical volumes globally (e.g., rising number of surgeries in emerging markets)
- Rising awareness of infection control protocols
- Expansion of OTC product lines in retail channels
- Government policies supporting hospital infection prevention
Risks and Challenges
- Competition from alcohol-based disinfectants and other antiseptics (e.g., chlorhexidine)
- Regulatory delays or restrictions
- Supply chain disruptions from raw material sourcing (iodine supply chain issues)
Opportunities and Strategic Outlook
- Diversification into new formulations, such as spray or foam versions
- Expanding into emerging markets via localized regulatory approvals
- Collaborations with hospital systems to establish standard disinfection protocols
- Promotion of Betadine’s use beyond traditional antisepsis, including COVID-19 related disinfection measures
Conclusion
Betadine's sales remain solid with steady regional growth; Asia-Pacific shows the most promise for expansion. The market is mature but continues to grow because of increasing healthcare needs, especially in developing countries. Plasticity in formulation and regulatory navigation will determine the pace of future expansion.
Key Takeaways
- The global antiseptic market was valued at USD 8 billion in 2022, with Betadine as a leading product.
- Asia-Pacific exhibits the fastest growth, driven by expanding healthcare infrastructure.
- Projected sales will reach approximately USD 12.03 billion by 2028, with a CAGR near 6%.
- Market growth is primarily fueled by rising surgical procedures, infection control measures, and OTC product availability.
- Competition from alcohol-based disinfectants and regulatory barriers pose ongoing challenges.
FAQs
Q1: How does Betadine compare with alcohol-based antiseptics?
A: Betadine offers a broader antimicrobial spectrum and is less irritating to skin than alcohol-based disinfectants but has a longer drying time.
Q2: What are the main regulatory hurdles for Betadine expansion?
A: Variations in approval processes, restrictions on off-label use, and supply chain issues for iodine can delay market entry.
Q3: How significant is the OTC market for Betadine?
A: OTC products constitute about 20% of Betadine's sales, predominantly in wound care and minor cuts in retail pharmacies.
Q4: Will new formulations impact Betadine’s market share?
A: Yes; innovations like spray or foam formulations can increase usability and expand consumer adoption.
Q5: What regions offer the most growth opportunities?
A: Asia-Pacific, Latin America, and Middle East & Africa due to rising healthcare spending and expanding infrastructure.
References
- Grand View Research, "Antiseptic Market Size & Trends," 2022.
- Mordor Intelligence, "Global Antiseptics Market Analysis," 2022.
- Johnson & Johnson, "Betadine Product Portfolio," 2022.
- World Health Organization, "Surgical Procedure Data," 2021.
- US Food and Drug Administration, "Antiseptic Drugs," 2022.
More… ↓
